But in one area, she hit a brick wall: attempting to force change at the California hospital where she believes her son contracted covid in December 2020. While seeking treatment for a bleeding cyst, Braden was surrounded for hours by coughing patients in the emergency room, Wilson said. Yet, she said, she has been unable to get the hospital to show her improvements it told her it made or get a lawyer to take her case.
“I was pretty shocked,” Wilson said. “There’s truly no recourse.”
Throughout the pandemic, have passed laws, declared emergency orders or activated state-of-emergency statutes that severely limited families’ ability to seek recourse for lapses in covid-related care.
Under such liability shields, legal advocates say, it’s nearly impossible to seek the legal accountability that can pry open information and drive systemic improvements to the infection-control practices that make hospitals safer for patients.
“Lawsuits are there for accountability and truth to be exposed,” said Kate Miceli, state affairs counsel for the , which advocates for plaintiff lawyers. “These laws are absolutely preventing that.”
A previous KHN investigation documented that more than 10,000 people tested positive for covid after they were hospitalized for something else in 2020. Yet many others, including Braden Wilson, are not counted in those numbers because they were discharged before testing positive. Still, the KHN findings are the only nationally publicly available data showing rates of patients who tested positive for covid after admission into individual U.S. hospitals.
Those who have lost a family member say hospitals need to be held more accountable.
“My mom is not like one of those people who would say тGo sue them,’” said Kim Crail, who believes her 79-year-old mom contracted covid during an eight-day stay at a hospital in Edgewood, Kentucky, because she tested positive less than 48 hours after leaving. “But she just wouldn’t want it to happen to anyone else.”
тYou Put Your Trust in the Hospital’
At age 89, Yan Keynigshteyn had begun to fade with dementia. But he was still living at home until he was admitted to Ronald Reagan UCLA Medical Center in Los Angeles for a urological condition, according to Terry Ayzman, his grandson.
Keynigshteyn, a Soviet Union emigrant who did not understand English, found himself in an unfamiliar place with masked caregivers. The hospital confined him to his bed, Ayzman said. He did not understand how to navigate the family’s Zoom calls and, eventually, stopped talking.
He was tested regularly for covid during his two-week-plus stay, Ayzman said. On Keynigshteyn’s way home in an ambulance, his doctor got test results showing he had tested positive for covid. It can take two to 14 days from exposure to covid for patients to start showing symptoms such as a fever, though the average is . His grandson believes that because Keynigshteyn was in the hospital for over two weeks before testing positive, he contracted covid at Ronald Reagan UCLA Medical Center.
As the ambulance doors opened and Keynigshteyn finally saw his wife and other family members, he smiled for the first time in weeks, Ayzman said. Then the crew slammed the doors shut and took him back to the hospital.

A few days later, Keynigshteyn died.
“You put your trust in the hospital and you get the short end of the stick,” Ayzman said. “It wasn’t supposed to be like that.”
Ayzman wanted to find out more from the hospital, but he said officials there refused to give him a copy of its investigation into his grandfather’s case, saying it was an internal matter and the results were inconclusive.
Hospital spokesperson Phil Hampton did not answer questions about Keynigshteyn. “UCLA Health’s overriding priority is the safety of patients, employees, visitors and volunteers,” he said, adding that the health system has been consistent with or exceeded infection-control protocols at the local, state and federal level throughout the pandemic.
Ayzman reached out to five lawyers, but he said none would take the case. He said they all told him courts were unsympathetic to cases against health care institutions at the time.
“I don’t believe that a state of emergency should give a license to hospitals to get away with things scot-free,” Ayzman said.

The Current State of Legal Play
The avalanche of liability shield legislation was pitched as a way to prevent a wave of lawsuits, Miceli said. But it created an “unreasonable standard” for patients and families, she said, since a state-of-emergency raises the bar for filing medical malpractice cases and already makes many lawyers hesitant to take such cases.
Almost every state put extra liability shield protections in place during the pandemic, Miceli said. Some of them broadly protected institutions such as hospitals, while others were more focused on shielding health care workers.
Corporate-backed groups, including the , the U.S. Chamber of Commerce , and the , helped pass a range of liability shield bills across the country through lobbying, working with state partners or drafting forms of model legislation, a KHN review has found.
William Melofchik, general counsel for NCOIL, said member legislators drafted their model bill because they felt it was important to guard against a never-ending wave of litigation and to be “better safe than sorry.”
Nathan Morris, vice president of legislative affairs for the Chamber’s Institute for Legal Reform, said his group’s work had influenced states across the country to implement what he called timely and effective protections for hospitals that were trying to do the right thing while working through a harrowing pandemic.
“Nothing that we advocated for would slam the courthouse door in the face of someone who had a claim that was clearly legitimate,” he said.
The other two organizations did not answer questions about their involvement in such work by deadline.

Joanne Doroshow, executive director of the at New York Law School, said such powerful corporate lobbying interests used the broader “health care heroes” moment to push through lawsuit protections for institutions like hospitals. She believes they will likely worsen .
“The fact that the hospitals were able to get immunity under these laws is pretty offensive and dangerous,” she said.
Some of the measures were time-limited or linked to public emergencies that have since expired, but, Miceli said, more than half of states still have some form of expanded liability laws and executive orders in place. Florida legislators are currently working to to mid-2023.
Doctors’ groups and hospital leaders say they in times of crisis.
“Liability protections can be incredibly important because they do encourage providers to continue working and to continue actually providing care in incredibly troubling emergency circumstances,” said , a deputy director of the Western Region Office for the Network for Public Health Law.
Akin Demehin, director of policy for the , said it’s important to remember the severe shortages in testing and personal protective equipment at the start of the pandemic. He added that the health care workforce faced tremendous strain as it had to juggle new roles amid personnel shortages, along with ever-evolving federal guidance and understanding of how the coronavirus spreads.
Piatt cautioned that appropriately calibrating liability shields is delicate work, as protections that are too broad can deprive patients of their ability to seek recourse.
Those wanting to learn more about how covid spreads within a U.S. hospital have few resources. Dr. , now an infectious diseases fellow at Stanford, and other researchers examined at Brigham and Women’s Hospital in Boston. But few hospitals have dug deep on the topic, he said, which could reflect the stretched-thin resources in hospitals or a fear of negative media coverage.
“There should be dialogue from the lessons learned,” Karan said.
тDo Not Put Anything in Writing’
Crail and Kelly Heeb lost their mother, , to covid early in 2021. The sisters believe she caught it during her more-than-weeklong stay at St. Elizabeth Edgewood Hospital outside Cincinnati following a hernia repair surgery.

They said she spent hours in an ER separated from other patients only by curtains and did not wear a mask in her patient room while she recovered. She was discharged from the hospital complaining about tightness in her chest, the sisters said. Within 24 hours, she spiked a fever. The next day, she was back in the ER, where she tested positive for covid on Christmas Eve 2020, they said. After a difficult bout with the virus, Terrell died Jan. 8.
When Crail attempted to file a complaint detailing their concerns, she said a hospital risk management employee told her: “тNo, do not put anything in writing.’”
Crail filed cursory paperwork anyway. She received the hospital’s conclusion in the mail in an envelope postmarked Dec. 1, more than seven months after the April 27 date typed at the top of the letterhead. The letter stated the St. Elizabeth Healthcare oversight committee determined it was “unable to substantiate” that their mother contracted covid in the hospital due to high community transmission rates, incubation timing and unreliable covid tests. The letter did note that despite the hospital system’s extensive protocols, “the risks of transmission will always exist.”
Guy Karrick, a spokesperson for the hospital, did not comment on the sisters’ specific case but said “we have not and would not tell any patient or family not to put their concerns in writing.” He added that the hospital has been following all federal and state guidelines to protect its patients.
Braden’s mom, Amanda Wilson, had far more dialogue with the hospital where she thinks her son got covid. But it still left her with doubts that she made an impact.
When her son was in the Adventist Health Simi Valley ER in December 2020 in a bed separated by curtains, they could hear staffers periodically reminding coughing patients around them to keep on their masks. She and Braden kept their own masks on for the vast majority of their several-hours-long stay, she said, but staffers in their bay didn’t always have their own masks pulled up.
Hospital spokesperson Alicia Gonzalez said staffers “track infections that may occur in our facilities and we have no verified infection of any patient or visitor of covid-19 in our facility,” adding that the hospital is “dedicated to serving our community and ensuring the safety of all who are cared for at our hospital.”

Wilson, a mathematician who works in the aerospace industry, expected the hospital to be able to show her evidence of some of the changes she discussed with hospital officials, including its president. For one, she hoped the staffers would get trained by a physician with direct experience treating the covid complication that made her son fatally ill, called MIS-C, or multisystem inflammatory syndrome. She also had hoped to see proof that the hospital installed no-touch faucets in the ER bathroom, which would help limit the spread of infections.
Gonzalez said that hospital executives listened to Wilson’s concerns and met with her on more than one occasion and that the hospital has improved its internal processes and procedures as it has learned about transmissibility and best practices.
But Wilson said they wouldn’t send her photos or let her see the changes for herself. The hospital declined to list or provide evidence of the changes to KHN as well.
“It made me more angry,” Wilson said. “Here I tried to make it better for people. I couldn’t make it better for Braden, but for people who’d come to this hospital т it is the only hospital in our town.”
She said she reached out to a lawyer, who told her there would be no way to prove how Braden caught covid. She had no other way to force more of a reckoning over her son’s death. So, she said, she has turned to other ways to “leave little pieces of him out in the world.”
This <a target="_blank" href="/health-industry/liability-laws-shield-hospitals-from-families-who-believe-loved-ones-contracted-covid-as-patients/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1423177&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>Patients were coughing as staffers wheeled the maskless soap opera star around the California hospital while treating her for injuries from a horseback fall in May 2020, Evans said.
She remembered they took her to a room to remove blood from her compressed lung where another maskless patient was also getting his lung drained. He was crying out that he didn’t want to die of covid.
No one had told her to wear a mask, she said. “It didn’t cross my mind, as I’m in a hospital where you’re supposed to be safe.”
Then, about a week into her hospital stay, she tested positive for covid-19. It left the 57-year-old hospitalized for a month, staring down more than $1 million in bills for treatment costs and suffering from debilitating long-haul symptoms, she said.
Hospitals, like Riverside, with high rates of covid patients who didn’t have the diagnosis when they were admitted have rarely been held accountable due to multiple gaps in government oversight, a KHN investigation has found.
While a federal reporting system closely tracks and other bugs, it doesn’t publicly report covid caught in individual hospitals.
Medicare officials, tapped by Congress decades ago to ensure quality care in hospitals, also discovered a gaping hole in their authority as covid spread through the nation. They private accreditors — which almost 90% of hospitals pay for oversight — to do targeted infection-control inspections. That means Riverside and nearly 4,200 other hospitals did not receive those specific covid-focused inspections, according to a , even though Medicare asked accreditors to do them in .
Seema Verma, former chief of Medicare and Medicaid under President Donald Trump, said government inspectors went into nearly every nursing home last year. That the same couldn’t be done for hospitals reveals a problem. “We didn’t have the authority,” she told KHN. “This is something to be corrected.”
KHN previously reported that at least 10,000 patients nationwide were diagnosed with covid in hospitals last year after being admitted for something else — a sure undercount of the infection’s spread inside hospitals, since that data analysis primarily includes Medicare patients 65 and older.
Nationally, 1.7% of Medicare inpatients were documented as having covid diagnosed after being admitted for another condition, according to data from April through September 2020 that hospitals reported to Medicare. CDIMD, a Nashville-based consulting and data analytics company, analyzed the data for KHN.
At Riverside Community Hospital, 4% of the covid Medicare patients were diagnosed after admission — more than double the national average. At 38 other hospitals, that rate was 5% or higher. All those hospitals are approved by private accreditors, and 29 of them hold “” from their accreditor.
To be sure, the data has limitations: It represents a difficult time in the pandemic, when protective gear and tests were scarce and vaccines were not yet available. And it could include community-acquired cases that were slow to show up. But hospital-employed medical coders decide whether a case of covid was present on admission based on doctors’ notes, and are trained to query doctors if it’s unclear. Some institutions fared better than others — while the American public was left in the dark.
Spurred by serious complaints, federal inspectors found infection-control issues in few of those 38 hospitals last year. In , inspectors reported that one hospital “failed to provide and maintain a sanitary environment resulting in the potential for the spread of infectious disease to 151 served by the facility.” In , inspectors found a hospital “failed to have an effective hospital-wide program for the surveillance and prevention” of covid.
KHN was able to find federal inspection reports documenting infection-control issues for eight of those 38 hospitals. The other 30 hospitals around the country, from Alabama to Arizona, had no publicly available federal records of infection-control problems in 2020.
KHN found that even when state inspectors in California assessed hospitals with high rates of covid diagnosed after admission, they identified few shortcomings.
“The American public thinks someone is watching over them,” said , co-founder of the Patient Safety Action Network, an advocacy group. “Generally they think someone’s in charge and going to make sure bad things don’t happen. Our oversight system in our country is so broken and so untrustworthy.”
The data shows that the problem has deadly consequences: About a fifth of the Medicare covid patients who were diagnosed after admission died. And it was costly as well. In California alone, the total hospital charges for such patients from April through December last year was over $845 million, according to an analysis done for KHN by the California Department of Health Care Access and Information.
The Centers for Disease Control and Prevention has pledged funding for increased infection-control efforts — but that money is not focused on tracking covid’s spread in hospitals. Instead, it will spend partly to support an existing tracking system for hospital-acquired pathogens such as and .
The CDC does not currently track hospital-acquired covid, nor does it plan to do so with the additional funding. That tracking is done by another part of the U.S. Department of Health and Human Services, according to , associate director for the CDC’s health care-associated infection-prevention programs. But it’s not made public on a hospital-by-hospital basis. HHS officials did not respond to questions.
The Scene at Riverside
In March 2020, Evans was alarmed by nonstop TV footage of covid deaths, so she and her husband locked down. They hadn’t been going out much, anyway, since losing their only child at the end of 2019 to another public health crisis — fentanyl.
At the time, concerns about covid were mounting among the staff at Riverside Community Hospital, a for-profit HCA Healthcare facility.
The hospital’s highly protective N95 masks had been pulled off the supply room shelves and put in a central office, according to Monique Hernandez, a shop steward for her union, Service Employees International Union Local 121RN. Only nurses who had patients getting aerosol-generating procedures such as intubation — which were believed at the time to spread the virus — could get one, she said.
She said that practice left the nurses on her unit with a difficult choice: either say you had a patient undergoing such procedures or risk getting sick.

Nurse unions were of the notion — now widely accepted — that covid is spread by minuscule particles that can linger in the air. Studies since the genetic fingerprint of the virus to show that covid has spread among workers or patients wearing surgical masks instead of more protective masks like N95s.
On April 22, 2020, Hernandez and other nurses joined a silent protest outside the hospital where they held up signs saying “PPE Over Profit.” By that time, the hospital had several staff clusters of infection, according to Hernandez, and she was tired of caregivers being at risk.
In a statement, Riverside spokesperson David Maxfield said the hospital’s top priority has been to protect staff “so they can best care for our patients.”
“Any suggestion otherwise ignores the extensive work, planning and training we have done to ensure the delivery of high-quality care during this pandemic,” he said.
In mid-May, Judi Evans’ husband coaxed her into going horseback riding — one of the few things that brought her joy after her son’s death. On her second day back in the saddle, she was thrown from her horse. She broke her collarbone and seven ribs, and her lung was compressed. She was taken to Riverside Community Hospital.

There, many of her nurses wore masks they had previously used, Evans recalled. Other staffers came in without any masks at all, she said. A few days in, she said, one of the doctors told her it’s crazy that the hospital was testing her for MRSA and other hospital infections but not covid.
Maxfield said that the hospital began enforcing a universal mask mandate for staff and visitors on March 31, 2020, and, “in line with CDC, patients were and are advised to wear masks when outside their room if tolerated.” He stressed “safety of our patients and colleagues has been our top priority.”
After about a week in the hospital, Evans said, she spiked a fever and begged for a covid test. It was positive. There is no way to know for certain where or how she got infected but she believes it was at Riverside. Covid infections can take two to 14 days from exposure to show symptoms like a fever, with the average being . According to , infection onset that occurs two days or more after admission could be “hospital-associated.”
Doctors told her they might have to amputate her legs when they began to swell uncontrollably, she said.
“It was like being in a horror film — one of those where everything that could go wrong does go wrong,” Evans said.
She left with over $1 million in bills from a month-long stay — and her legs, thankfully. She said she still suffers from long-covid symptoms and is haunted by the screams of fellow patients in the covid ward.

By the end of that year, Riverside Community Hospital would report that 58 of its 1,649 covid patients were diagnosed with the virus after admission, according to state data that covers all payers from April to December.
That’s nearly three times as high as the California average for covid cases not present on admission, according to the analysis for KHN by California health data officials.
“Based on contact tracing, outlined by the CDC and other infectious disease experts, there is no evidence to suggest the risk of transmission at our hospital is different than what you would find at other hospitals,” Maxfield said.
in August by the SEIU-United Healthcare Workers West on behalf of the daughter of a lab assistant who died of covid and other hospital staffers says the hospital forced employees to work without adequate protective gear and while sick and “highly contagious.”
The hospital “created an unnecessarily dangerous work environment,” the lawsuit claims, “which in turn has created dangerous conditions for patients” and a “public nuisance.”
Attorneys for Riverside Community Hospital are . “This lawsuit is an attempt for the union to gain publicity, and we have filed a motion to end it,” said Maxfield, the hospital spokesperson.
The hospital’s lawyers have said the plaintiffs got covid during a spike in local cases and are only speculating that they contracted the virus at the hospital, according to records filed in Riverside County Superior Court.
They also said in legal filings that the court should not step into the place of “government agencies who oversee healthcare and workplace safety” and “handled the response to the pandemic.”

‘A Shortcoming in the Oversight System’
, Congress tasked Medicare with ensuring safe, quality care in U.S. hospitals by building in routine government inspections. However, hospitals can opt to of dollars per year to nongovernmental accreditors entrusted by CMS to certify the hospitals as safe. So 90% do just that.
But these accrediting agencies — including the Joint Commission, which certified Riverside — are private organizations. Thus they are not required to follow CMS’ directives, including the request in a urging the accrediting agencies to execute targeted infection-control surveys aimed at preparing hospitals for covid’s onslaught.
And so they didn’t send staffers to survey hospitals for the specialized infection-control inspections in 2020, according to a June 2021 .
Riverside, despite allegations of lax practices, holds from the Joint Commission, which the hospital on-site in May 2018 before going in on Nov. 19 this year.
The inspector general’s office urged CMS to pursue the authority to require special surveys in a health emergency — lest it lose control of its mission to keep hospitals safe.
“CMS could not ensure that accredited hospitals would continue to provide quality care and operate safely during the COVID-19 emergency,” and could not ensure it going forward, said.
“We’re telling CMS to do their job,” the report’s author, Assistant Regional Inspector General Calvin Jones, said in an interview. “The covid experience really showed a shortcoming in the oversight system.”
CMS spokesperson Raymond Thorn said the agency agrees with the report’s recommendation and will work on a regulation after the public health emergency ends.
Accrediting agencies, however, pushed back on the inspector general’s findings. Among them: DNV Healthcare USA Inc. Its director of accreditation, Troy McCann, said there was not a gap in oversight. Although he said travel restrictions limited accreditors ability to fly across state lines, his group continued its annual reviews after May 2020 and incorporated the special focus on infection control into them. “We have a strong emphasis, always, on safety, infection control and emergency preparedness, which has left our hospitals stronger,” McCann said.
Angela FitzSimmons, spokesperson for the Accreditation Commission for Health Care, said that the accrediting organization’s surveys typically focus on infection control, and the group worked during the pandemic to prioritize hospitals with prior issues in the area of infection prevention.
“We did not deem it necessary to add random surveys that would occur at a cost to the hospital without just cause,” FitzSimmons said.
Maureen Lyons, a spokesperson for the Joint Commission, told KHN that, after evaluating CMS guidance, the nonprofit group decided it would incorporate the infection-control surveys into its surveys done every three years and, in the meantime, provide hospitals with the latest federal guidance on covid.
“Hospitals were operating in extremis. Thus, we collaborated closely with CMS to determine optimal strategies during this time of emergency,” she said.
The Joint Commission cited safety issues for its inspectors, who travel to the hospitals and need proper protective equipment that was running low at the time, as part of the reason for its decision.
Verma, the CMS administrator at the time, pushed back on accreditors’ travel safety concerns, saying that “narrative doesn’t quite fit because the state and CMS surveyors were going into nursing homes.”
Though Verma cautioned that hospitals were overwhelmed by the crush of covid patients, “doing these inspections may have helped hospitals bolster their infection-control practices,” she said. “Without these surveys, we really have no way of knowing.”
‘Immediate Jeopardy’
Medicare inspectors can go into a privately accredited hospital after they get a serious complaint. They found alarming circumstances when they visited some of the hospitals with high rates of covid diagnosed after a patient was admitted for another concern last year.
At Levindale Hebrew Geriatric Center and Hospital in Baltimore, says “systemic failures left the hospital and all of its patients, staff, and visitors vulnerable to harm and possible death from COVID-19.”
In response, hospital spokesperson Sharon Boston said that “we have seen a large decrease in the spread of the virus at Levindale.”
Inspectors had declared a state of “immediate jeopardy” after they investigated a complaint and discovered an outbreak that began in April and continued through the beginning of July, with more than 120 patients and employees infected with covid. And in a unit for those with Alzheimer’s and other conditions, 20% of the 55 patients who had covid died.
The hospital moved patients whose roommates tested positive for covid to other shared rooms, “potentially exposing their new roommate,” the inspection report said. Boston said that was an “isolated” incident and the situation was corrected the next day, with new policies put in place.
The Medicare data analyzed exclusively for KHN shows that 52 of Levindale’s 64 covid hospital patients, or 81%, were diagnosed with covid after admission from April to September 2020. Boston cited different numbers over a different time period: Of 67 covid patients, 64 had what she called “hospital-acquired” covid from March to June 2020. That would be nearly 96%.
The hospital shares space with a nursing home, though, so KHN did not group it with the general short-term acute-care hospitals as part of the analysis. Levindale’s last Joint Commission was in December 2018, resulting in The Gold Seal of Approval. It had not had its once-every-three-years survey as of Dec. 10, 2021, according to .
Boston said Levindale “quickly addressed” the issues that Medicare inspectors cited, increasing patient testing and more recently mandating staff vaccines. Since December 2020, Boston said, the facility has not had a covid patient die.
At the state level, hospital inspectors in California found few problems to cite even at hospitals where 5% or more patients were diagnosed with covid after they were admitted for another concern. Fifty-three complaints about such hospitals went to the Department of Public Health from April until the end of 2020. Only three of those complaints resulted in a finding of deficiency that facility was expected to fix.
CDPH did not respond to requests for comment.

A New Chapter
Things are better now at Riverside Community Hospital, Hernandez said. She is pleased with the current safety practices, including more protective gear and HEPA filters for covid patients’ rooms. For Hernandez, though, it all comes too late now.
“We laugh at it,” she said, “but it hurts your soul.”
Evans said she was able to negotiate her $1 million-plus hospital bills down to roughly $70,000.
Her covid aftereffects have been ongoing — she said she stopped gasping for air and reaching for her at-home oxygen tank only a few months ago. She still hasn’t been able to return to work full time, she said.
For the past year, her husband would wake up in the middle of the night to check whether her oxygen levels were dipping. Terrified of losing her, he’d slip an oxygen mask on her face, she said.
“I would walk 1,000 miles to go to another hospital,” Evans said, if she could do it all over again. “I would never step foot in that hospital again.”
Methodology
KHN requested custom analyses of Medicare, California and Florida inpatient hospital data to examine the number of covid-19 cases diagnosed after a patient’s admission.
The Medicare and Medicare Advantage data, which includes patients who are 65 and older, is from the Centers for Medicare & Medicaid Services’ Medicare Provider Analysis and Review (MedPAR) file and was analyzed by CDIMD, a Nashville-based medical code consulting and data analytics firm. The data is from April 1 through Sept. 30, 2020. The data for the fourth quarter of 2020 was not yet available.
The data shows the number of inpatient Medicare hospital stays in the U.S., including the number of people diagnosed with covid-19 and the number of admissions for which the covid diagnosis was not “present on admission.” CMS considers some medical conditions that are not “present on admission” to be hospital-acquired, according . The data is for general acute-care hospitals, which may include a psychiatric floor, and not for other hospitals such as those in the Department of Veterans Affairs system or stand-alone psychiatric hospitals.
KHN requested a similar analysis from California’s Department of Health Care Access and Information of its hospital inpatient data. That data was from April 1 through Dec. 31, 2020, and covered patients of all ages and payer types and, in general, private psychiatric and long-term acute-care hospitals. Etienne Pracht, a University of South Florida researcher, provided the number of Florida covid patients who did not have the virus upon hospital admission for all ages and payer types at general and psychiatric hospitals from April 1 through Dec. 31, 2020. KHN subtracted the number of Medicare patients in the MedPAR data from the Florida and California datasets so they would not be counted twice.
To calculate the rate of hospitalized Medicare patients who tested positive for covid — and died — KHN relied on the MedPAR data for April through September. That data includes records for 6,629 seniors, 1,409 of whom, or 21%, died. California data for all ages and payer types from April through December shows a similar rate: Of 2,115 diagnosed with covid-19 after hospital admission, 435, or 21%, died. The MedPAR data was also used to calculate the national rate of 1.7%, with 6,629 of 394,939 covid patients diagnosed with the virus whose infections were deemed not present on admission, according to the CDIMD analysis of data that hospitals report to Medicare. It was also used to calculate which entities licensed as short-term acute care hospitals had 5% or more of their covid cases diagnosed within the hospital. As stated in the story, Levindale Hebrew Geriatric Center and Hospital in Baltimore was not included in that list of 38 because it shares space with a nursing home and had fewer than 500 total discharges.
Data that hospitals submit to Medicare on whether an inpatient hospital diagnosis was “present on admission” is for payment determinations and is intended to incentivize hospitals to prevent infections during hospital care. The federal Agency for Healthcare Research and Quality also uses the data to “assist in identifying quality of care issues.”
Whether covid-19 is acquired in a hospital or in the community is measured in different ways. Some nations assume the virus is hospital-acquired if it is diagnosed seven or more days after admission, while counts cases only after 14 days.
Hospitals’ medical coders who examined patient records for the data analyzed for this KHN report focus on each physician’s admission, progress and discharge notes to determine whether covid was “present on admission.” They do not have a set number of days they look for and are trained to query physicians if the case is unclear, according to Sue Bowman, senior director of coding policy and compliance at the American Health Information Management Association.
KHN tallied the cases in which covid-19 was logged in the data as not “present on admission” to the hospital. Some covid cases are coded as “U” for having insufficient documentation to make a determination. Since Medicare and AHRQ consider the “U” to be an “N” (or not present on admission) for the purposes of and , KHN chose to count those cases in the grand total.
In 409 of 6,629 Medicare cases and in 70 of 2,185 California cases, the “present on admission” indicator was “U.” The Florida data did not include patients whose “present on admission” indicator was “U.” Medical coders have another code, “W,” for “clinically undetermined” cases, which consider a condition present on admission for billing or quality measures. Medical coders use the “U” (leaning toward “not present on admission”) and “W” (leaning toward “present on admission”) when there is some uncertainty about the case. KHN did not count “W” cases.
The Medicare MedPAR data includes about 2,500 U.S. hospitals that had at least a dozen covid-19 cases from April through September 2020. Of those, 1,070 reported no cases of covid diagnosed after admission for other conditions in the Medicare records. Data was suppressed due to privacy reasons for about 1,300 hospitals that had between one and 11 of such covid cases. There were 126 hospitals reporting 12 or more cases of covid that were “not present on admission” or unknown. For those, we divided the number of cases diagnosed after admission by the total number of patients with covid to arrive at the rate, as is standard in health care.
Inspection and Accreditation Analysis
To evaluate which of the 38 hospitals detailed above had federal inspection reports documenting infection-control issues, KHN searched CMS’ , which detail deficiencies for each hospital for 2020. For surveys listed online as “not available,” KHN requested and obtained them from CMS. KHN further asked CMS to double-check the remaining hospitals for any inspection reports that weren’t posted online. KHN also checked the Association of Health Care Journalists’ database for each of the 38 hospitals for any additional reports, as well as CMS’ site.
To check that each of these hospitals was accredited, KHN looked up each hospital run by the Joint Commission and reached out to the accreditors DNV Healthcare USA Inc. and the Accreditation Commission for Health Care.
To tabulate infection-control complaints for hospitals at the state level in California, KHN used data available through the California Department of Public Health’s . KHN searched the database for the hospitals that had higher than 5% of covid patients being diagnosed after admission, according to the California data, and tallied all complaints and deficiencies found involving infection control from April to December 2020.
This <a target="_blank" href="/health-industry/as-patients-fell-ill-with-covid-inside-hospitals-government-oversight-fell-short/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1424556&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>They left with covid-19 — if they left at all.
More than 10,000 patients were diagnosed with covid in a U.S. hospital last year after they were admitted for something else, according to federal and state records analyzed exclusively for KHN. The number is certainly an undercount, since it includes mostly patients 65 and older, plus California and Florida patients of all ages.
Yet in the scheme of things that can go wrong in a hospital, it is catastrophic: About 21% of the patients who contracted covid in the hospital from April to September last year died, the data shows. In contrast, nearly 8% of other Medicare patients died in the hospital at the time.

Steven Johnson, 66, was expecting to get an infection cut out of his hip flesh and bone at Blake Medical Center in Bradenton, Florida, last November. The retired pharmacist had survived colon cancer and was meticulous to avoid contracting covid. He could not have known that, from April through September, 8% of that hospital’s Medicare covid patients were diagnosed with the virus after they were admitted for another concern.
Johnson had tested negative for covid two days before he was admitted. After 13 days in the hospital, he tested positive, said his wife, Cindy Johnson, also a retired pharmacist.
Soon he was struggling to clear a glue-like phlegm from his lungs. A medical team could hardly control his pain. They prompted Cindy to share his final wishes. She asked: “Honey, do you want to be intubated?” He responded with an emphatic “no.” He died three days later.
After her husband tested positive, Cindy Johnson, trained in contact tracing, quickly got a covid test. She tested negative. Then she thought about the large number of hospital staffers flowing into and out of his room — where he was often unmasked — and suspected a staff member had infected him. That the hospital, part of the HCA Healthcare chain, still has not mandated staff vaccinations is “appalling,” she said.
“I’m furious,” she said.
“How can they say on their website,” she asked, “that the safety precautions ‘we’ve put into place make our facilities among the safest possible places to receive healthcare at this time’?”
Blake Medical Center spokesperson Lisa Kirkland said the hospital is “strongly encouraging vaccination” and noted that it follows Centers for Disease Control and Prevention and federal and state guidelines to protect patients. President Joe Biden has called for all hospital employees to be vaccinated, but the requirement could face resistance in , including Florida, that have banned vaccine mandates.

Overall, the rate of in-hospital spread among Medicare and other patients was lower than in other countries, including the United Kingdom, which makes such and openly discusses it. On average, about 1.7% of U.S. hospitalized covid patients were diagnosed with the virus in U.S. hospitals, according to an analysis of Medicare records from April 1 to Sept. 30, 2020, provided by Dr. James Kennedy, founder of CDIMD, a Nashville-based consulting and data analytics company.
Yet the rate of infection was far higher in 38 hospitals where 5% or more of the Medicare covid cases were documented as hospital-acquired. The data is from a challenging stretch last year when protective gear was in short supply and tests were scarce or slow to produce results. The Medicare data for the fourth quarter of 2020 and this year isn’t available yet, and the state data reflects April 1 through Dec. 31, 2020.
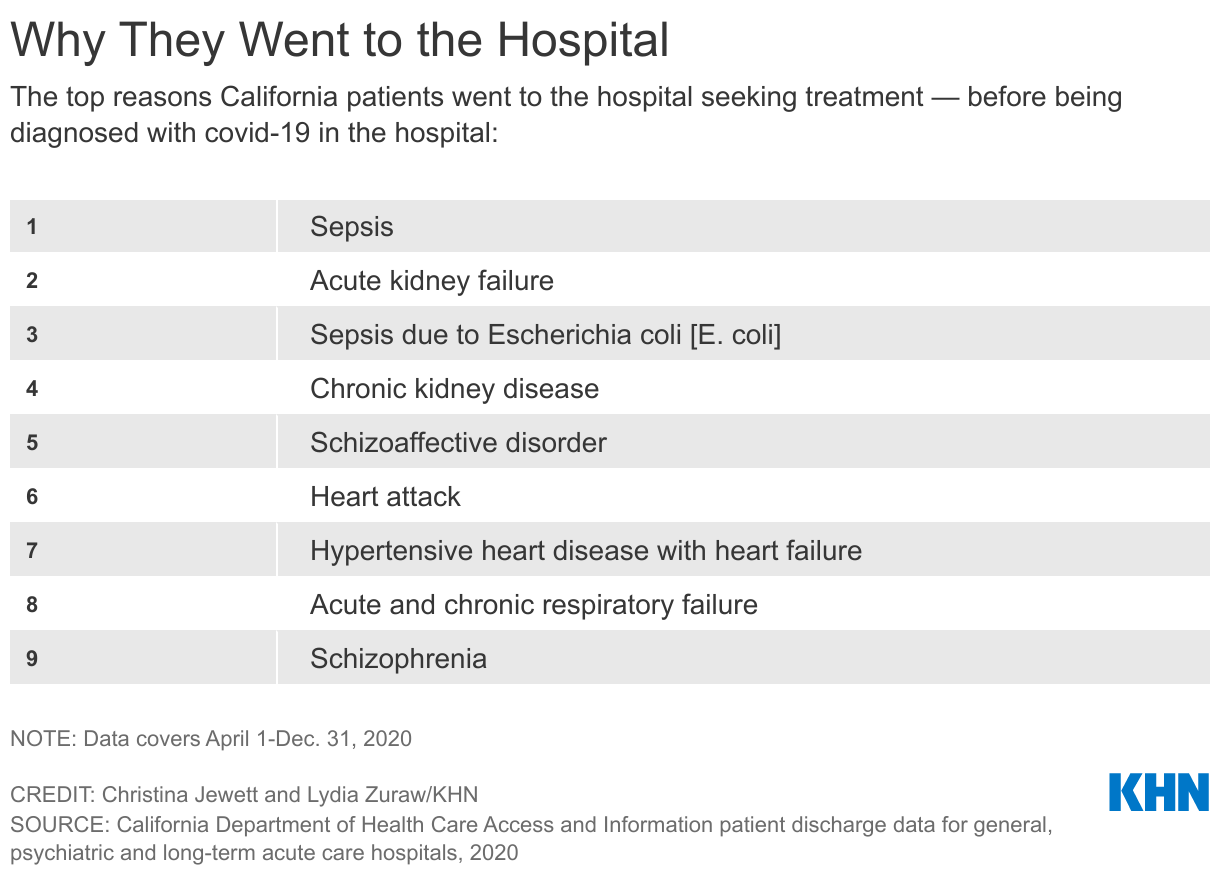
A KHN review of work-safety records, medical literature and interviews with staff at high-spread hospitals points to why the virus took hold: Hospital leaders were slow to appreciate its airborne nature, which made coughing patients hazardous to roommates and staff members, who often wore less-protective surgical masks instead of N95s. Hospitals failed to test every admitted patient, enabled by CDC guidance that leaves such testing to the “discretion of .” Management often failed to inform workers when they’d been exposed to covid and so were at risk of spreading it themselves.
Spread among patients and staffers seemed to go hand in hand. At Beaumont Hospital, Taylor, in Michigan, 139 employee covid infections were logged between April 6 to Oct. 20 last year, a hospital shows. Nearly 7% of the Medicare patients with covid tested positive after they were admitted to that hospital for something else, the federal data shows. A hospital spokesperson said tests were not available to screen all patients last year, resulting in some late diagnoses. He said all incoming patients are tested now.
Tracking covid inside health facilities is no new task to federal officials, who publicly report new staff and resident for each U.S. nursing home. Yet the Department of Health and Human Services on covid’s spread in hospitals only on a statewide basis, so patients are in the dark about which facilities have cases.
KHN commissioned analyses of hospital billing records, which are also used more broadly to spot various infections. For covid, the data has limitations. It can pick up some community-acquired cases that were slow to show up, as it can take two to 14 days from exposure to the virus for symptoms to appear, with the average being . The records do not account for cases picked up in an emergency room or diagnosed after a hospital patient was discharged.

Linda Moore, 71, tested positive at least 15 days into a hospital stay for spinal surgery, according to her daughter Trisha Tavolazzi. Her mother was at Havasu Regional Medical Center in Lake Havasu City, Arizona, which did not have a higher-than-average rate of internal spread last summer.
The hospital implemented “rigorous health and safety protocols to protect all of our patients” during the pandemic, said hospital spokesperson Corey Santoriello, who would not comment on Moore’s case, citing privacy laws.
Moore was airlifted to another hospital, where her condition only declined further, her daughter said. After the ventilator was removed, she clung to life fitfully for 5½ hours, as her daughter prayed for her mother to find her way to heaven.
“I asked her mom and her dad and her family and prayed to God, ‘Please just come show her the way,’” Tavolazzi said. “I relive it every day.”
When Tavolazzi sought answers from the hospital about where her mom got the virus, she said, she got none: “No one ever called me back.”
Two Negative Covid Tests, Then ‘Patient Zero’
As the second surge of covid subsided last September, doctors from the prestigious Brigham and Women’s Hospital published a reassuring : With careful infection control, only two of 697 covid patients acquired the virus within the Boston hospital. That is about 0.3% of patients ― about six times lower than the overall Medicare rate. Brigham tested every patient it admitted, exceeding CDC recommendations. It was transparent and open about safety concerns.
But the study, published in the high-profile JAMA Network Open journal, conveyed the wrong message, according to Dr. Manoj Jain, an infectious-disease physician and adjunct professor at the Rollins School of Public Health at Emory University. Covid was spreading in hospitals, he said, and the study buried “the problem under the rug.”
Before the virtual ink on the study was dry, the virus began a stealthy streak through the elite hospital. It slipped in with a patient who tested negative twice ― but turned out to be positive. She was “patient zero” in an outbreak affecting 38 staffers and 14 patients, according in Annals of Internal Medicine initially published Feb. 9.
That ’s authors sequenced the genome of the virus to confirm which cases were related ― and precisely how it traveled through the hospital.
As patients were moved from room to room in the early days of the outbreak, covid spread among roommates 8 out of 9 times, likely through aerosol transmission, the study says. A survey of staff members revealed that those caring for coughing patients were more likely to get sick.
The virus also appeared to have breached the CDC-OK’d protective gear. Two staff members who had close patient contact while wearing a surgical mask and face shield still wound up infected. The findings suggested that more-protective N95 respirators could help safeguard staff.
Brigham and Women’s now tests every patient upon admission and again soon after. Nurses are encouraged to test again if they see a subtle sign of covid, said Dr. Erica Shenoy, associate chief of the Infection Control Unit at Massachusetts General Hospital, who helped craft policy at Brigham.
She said nurses and environmental services workers are at the table for policymaking: “I personally make it a point to say, ‘Tell me what you’re thinking,’” Shenoy said. “’There’s no retribution because we need to know.’”
CDC guidelines, though, left wide latitude on protective gear and testing. To this day, Shenoy said, hospitals employ a wide range of policies.
The CDC said in a statement that its guidelines “provide a comprehensive and layered approach to preventing transmission of SARS-CoV-2 in healthcare settings,” and include testing patients with “even mild symptoms” or recent exposure to someone with covid.
Infection control policies are rarely apparent to patients or visitors, beyond whether they’re asked to wear a mask. But reviews of public records and interviews with more than a dozen people show that at hospitals with high rates of covid spread, staff members were often alarmed by the lack of safety practices.

Nurses Sound the Alarm on Covid Spread
As covid crept into Florida in spring 2020, nurse Victoria Holland clashed with managers at Blake Medical Center in Bradenton, where Steven Johnson died.
She said managers suspended her early in the pandemic after taking part in a protest and “having a hissy fit” when she was denied a new N95 respirator before an “aerosol-generating” procedure. The CDC warns that such procedures can spread the virus through the air. Before the pandemic, nurses were trained to dispose of an N95 after each patient encounter.
When the suspension was over, Holland said, she felt unsafe. “They told us nothing,” she said. “It was all a little whisper between the doctors. You had potential covids and you’d get a little surgical mask because [they didn’t] want to waste” an N95 unless they knew the patient was positive.
Holland said she quit in mid-April. Her nursing colleagues lodged a complaint with the Occupational Safety and Health Administration in late June alleging that staff “working around possible Covid-19 positive cases” had been denied PPE. Staff members protested outside the hospital and filed another OSHA complaint that said the hospital was allowing covid-exposed employees to keep working.
Kirkland, the Blake spokesperson, said the hospital responded to OSHA and “no deficiencies were identified.”
The Medicare analysis shows that 22 of 273 patients with covid, or 8%, were diagnosed with the virus after they were admitted to Blake. That’s about five times as high as the national average.
Kirkland said “there is no standard way for measuring COVID-19 hospital-associated transmissions” and “there is no evidence to suggest the risk of transmission at Blake Medical Center is different than what you would find at other hospitals.”
In Washington, D.C., 34 Medicare covid patients contracted the virus at MedStar Washington Hospital Center, or nearly 6% of its total, the analysis shows.
Unhappy with the safety practices ― which included gas sterilization and reuse of N95s — National Nurses United members on the hospital lawn in July 2020. At the protest, nurse Zoe Bendixen said one nurse had died of the virus and 50 had gotten sick: “[Nurses] can become a source for spreading the disease to other patients, co-workers and family members.”
Nurse Yuhana Gidey said she caught covid after treating a patient who turned out to be infected. Another nurse ― not managers doing contact tracing ― told her she’d been exposed, she said.
Nurse Kimberly Walsh said in an interview there was an outbreak in a geriatric unit where she worked in September 2020. She said management blamed nurses for bringing the virus into the unit. But Walsh pointed to another problem: The hospital wasn’t covid-testing patients coming in from nursing homes, where spread was rampant last year.
MedStar declined a request for an interview about its infection control practices and did not respond to specific questions.
While hospitals must track and rates of persistent infections like C. diff, antibiotic-resistant staph and surgical site infections, similar hospital-acquired covid rates are not reported.
KHN examined a different source of data that Congress required about “hospital-acquired conditions.” The Medicare data, which notes whether each covid case was “present on admission” or not, becomes available months after a hospitalization in obscure files that require a data-use agreement typically granted to researchers. KHN counted cases, as federal officials do, in some instances in which the documentation is deemed insufficient to categorize a case (see data methodology, below).
For this data, whether to deem a covid case hospital-acquired lies with medical coders who review doctors’ notes and discharge summaries and ask doctors questions if the status is unclear, said Sue Bowman, senior director of coding policy and compliance at American Health Information Management Association.
She said medical coders are aware that the data is used for hospital quality measures and would be careful to review the contact tracing or other information in the medical record.
If a case was in the data KHN used, “that would mean it was acquired during the hospital stay either from a health care worker or another patient or maybe if a hospital allowed visitors, from a visitor,” Bowman said. “That would be a fair interpretation of the data.”
The high death rate for those diagnosed with covid during a hospital stay — about 21% — mirrors the death rate for other Medicare covid patients last year, when doctors had few proven methods to help patients. It also highlights the hazard unvaccinated staffers pose to patients, said Jain, the infectious-disease doctor. The American Hospital Association estimates that about 42% of U.S. hospitals have mandated that all staff members be vaccinated.
“We don’t need [unvaccinated staff] to be a threat to patients,” Jain said. “[Hospital] administration is too afraid to push the nursing staff, and the general public is clueless at what a threat a non-vaccinated person poses to a vulnerable population.”

Cindy Johnson said the hospital where she believes her husband contracted covid faced minimal scrutiny in a state , even after she said she reported that he caught covid there. She explored suing, but an attorney told her it would be nearly impossible to win such a case. A 2021 requires proof of “at least gross negligence” to prevail in court.
Johnson did ask a doctor who sees patients at the hospital for this: Please take down the big “OPEN & SAFE” sign outside.
Within days, the sign was gone.
KHN Midwest correspondent Lauren Weber contributed to this report.
Methodology
KHN requested custom analyses of Medicare, California and Florida inpatient hospital data to examine the number of covid-19 cases diagnosed after a patient’s admission.
The Medicare and Medicare Advantage data, which includes patients who are mostly 65 or older, is from the Medicare Provider Analysis and Review (MedPAR) file and was analyzed by CDIMD, a Nashville-based medical code consulting and data analytics firm. The data is from April 1 through Sept. 30, 2020. The data for the fourth quarter of 2020 is not yet available.
That data shows the number of inpatient Medicare hospital stays in the U.S., including the number of people diagnosed with covid and the number of admissions for which the covid diagnosis was not “present on admission.” A condition not “present on admission” is presumed to be hospital-acquired. The data is for general acute-care hospitals, which may include a psychiatric floor, and not for other hospitals such as Veterans Affairs or stand-alone psychiatric hospitals.
KHN requested a similar analysis from California’s Department of Health Care Access and Information of its hospital inpatient data. That data was from April 1 through Dec. 31, 2020, and covered patients of all ages and payer types and in general, private psychiatric and long-term acute-care hospitals. Etienne Pracht, a University of South Florida researcher, provided the number of Florida covid patients who did not have the virus upon hospital admission for all ages at general and psychiatric hospitals from April 1 through Dec. 31, 2020. KHN subtracted the number of Medicare patients in the MedPAR data from the Florida and California all-payer datasets so they would not be counted twice.
To calculate the rate of Medicare patients who got covid or died, KHN relied on the MedPAR data for April through September. That data includes records for 6,629 seniors, 1,409 of whom, or 21%, died. California data for all ages and payer types from April through December shows a similar rate: Of 2,115 who contracted covid after hospital admission, 435, or 21%, died. The MedPAR data was also used to calculate the national nosocomial covid rate of 1.7%, with 6,629 of 394,939 covid patients diagnosed with the virus that was deemed not present on admission.
Data on whether an inpatient hospital diagnosis was present on admission is for payment determinations and is intended to incentivize hospitals to prevent infections acquired during hospital care. It is the U.S. Agency for Healthcare Research and Quality to “assist in identifying quality of care issues.”
Whether covid is acquired in a hospital or in the community is measured in different ways. Some nations assume the virus is hospital-acquired if it is diagnosed seven or more days after admission, while statewide counts cases only after 14 days.
Medical coders who examine medical records for this inpatient billing data focus on the physician’s admission, progress and discharge notes to determine whether covid was present on admission. They do not have a set number of days they look for and are trained to query physicians if the case is unclear, according to Sue Bowman, senior director of coding policy and compliance at the American Health Information Management Association.
KHN tallied the cases in which covid was logged in the data as not “present on admission” to the hospital. Some covid cases are coded as “U” for having insufficient documentation to make a determination. Since Medicare and AHRQ consider the “U” to be an “N” (or not present on admission) for the purposes of and , KHN chose to count those cases in the grand total.
In 409 of 6,629 Medicare cases and in 70 of 2,185 California cases, the “present on admission” indicator was “U.” The Florida data did not include patients whose “present on admission” indicator was “U.” Medical coders have another code, “W,” for “clinically undetermined” cases, which consider a condition present on admission for billing or quality measures. Medical coders use the “U” (leaning toward “not present on admission”) and “W” (leaning toward “present on admission”) when there is some uncertainty about the case.
The Medicare MedPAR data includes about 2,500 U.S. hospitals that had at least a dozen covid cases from April through September 2020. Of those, 1,070 reported no cases of hospital-acquired covid in the Medicare records. Data was suppressed for privacy reasons for about 1,300 hospitals that had between one and 11 hospital-acquired covid cases. There were 126 hospitals reporting 12 or more cases of covid that were not present on admission or unknown. For those, we divided the number of hospital-acquired cases by the total number of patients with covid to arrive at the rate of hospital-acquired cases, as is standard in health care.
This <a target="_blank" href="/health-industry/hospital-acquired-covid-nosocomial-cases-data-analysis/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1399366&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>The , led by Democratic Reps. Raja Krishnamoorthi and Katie Porter, focuses on conflicts of interest held by members of a committee currently reviewing a life-or-death matter: U.S. organ donation and distribution policy. A panel member recently resigned after accepting a consulting job that apparently created a conflict of interest. House members are asking NASEM to provide conflict-of-interest disclosure forms for all members of the committee.
Members of Congress want to ensure that reports from the national academies, chartered in 1863 to provide Congress with expert scientific advice, are widely accepted as independent and free from special-interest or financial influence.
Starting Sept. 7, NASEM is requiring committee members to disclose relevant financial relationships for the past five years, going beyond its recent policy of asking for details about current conflicts, spokesperson Dana Korsen said.
The institution conflict-of-interest policies are meant to protect it from “a situation where others could reasonably question, and perhaps discount or dismiss, the work of the committee simply because of the existence of such conflicting interests.”
This summer, the national academies declined to provide KHN with conflict-of-interest forms completed by committee members studying the packaging of liquid drugs, which often go to waste т along with millions of taxpayer dollars paid to pharmaceutical firms for them. The final report declared conflicts for two members, but not for two others who had extensive and recent financial ties to drug companies. At that time, the organization said those committee members had no “current” conflicts while they drafted the report, mostly in 2020.
A review of public records showed that one member reported for medical journals in 2021 and 2020 that he had consulted for a dozen pharmaceutical firms. Another member had been paid about $1.4 million as a pharmaceutical company board member in 2019, according to a Securities and Exchange Commission filing. Both told KHN they had fully reported their financial relationships to the national academies.
In addition, NASEM itself had disclosed in obscure treasurer that pharmaceutical companies т many with a direct interest in drug-waste policy т had given the nonprofit organization at least $10 million in donations since 2015.
The final NASEM drug-waste in February concluded that drug companies should not refund taxpayers for the cost of wasted medications packaged in “-sized” vials that contain more drugs than can be used. What’s more, Medicare should not track the cost of the wasted drugs, the report concluded.
On Friday, on the resignation of a former organ transplant doctor from a NASEM committee reviewing organ transplant policies. Dr. Yolanda Becker resigned from the organ donation committee after the Post asked her and NASEM questions about a possible conflict of interest.
Becker’s resignation comes amid continued scrutiny of the U.S. organ transplant system, which is overseen by the United Network for Organ Sharing, or UNOS, a nonprofit federal contractor.
Critics, including members of Congress, have questioned the performance of many of the nation’s 57 organ procurement organizations, or OPOs, which hold federal charters that guarantee their monopolies to collect and distribute organs in specific geographic areas of the United States.
“The organ transplant industry has long been a haven for fraud, waste, and abuse,” Rep. Porter said in a statement to KHN. “I’m grateful NASEM is also working to hold OPOs accountable, but I’m concerned that potential conflicts of interest could cloud their judgement.”
Jennifer Erickson, a senior fellow and director of the organs initiative at the nonpartisan Federation of American Scientists, raised questions about conflicts of interest with NASEM during a
“Disclosure is critical. The public deserves to know about conflicts of interest and undisclosed payments,” she said. “A good start would be for all members of this committee to publicly disclose their business relationships related to organ contractors, [organ] tissue businesses and trade associations so that the public can be aware.”
The Trump administration in an effort to boost the numbers of organs transplanted by OPOs by more than 7,000 a year. Nearly 107,000 people in the United States are awaiting organs, and dozens die each day for lack of a transplant. About 39,000 organs were transplanted from donors in the U.S. last year.
In May, chaired by Krishnamoorthi held a hearing on problems in the organ transplant system, including issues revealed by reporting from KHN and Reveal that found that donated organs т mostly kidneys т are repeatedly lost or damaged when shipped via commercial flights. From 2014 to 2019, nearly 170 organs failed to be transplanted and almost 370 endured “near misses” with delays of two hours or more, jeopardizing their usefulness for ailing patients.
Reps. Krishnamoorthi and Porter have asked NASEM to provide them with an explanation of whether it plans to disclose any committee conflicts in the forthcoming organ report. They also requested any record of donations to NASEM from organ procurement organizations or related businesses or associations.
бюЙѓхњДЋУНвюl Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFFтan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/health-care-costs/national-academies-conflict-of-interest-congress-cites-khn-investigation-drug-waste/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1378136&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>So Congress turned to the prestigious National Academies of Sciences, Engineering and Medicine for advice, given its reputation for “, objective reports” on such matters. The national academies’ influential report, released in February, struck physicians who’ve tracked the issue as distinctly friendly to Big Pharma. It advised against an effort to recoup millions for the discarded drugs. It concluded that Medicare should stop tracking the cost of the drug waste altogether.
Yet the report left out a few key facts, a KHN investigation has found.
Among them: One committee member was paid $1.4 million to serve on the board of a pharmaceutical corporation in 2019 and in 2020 joined the board of a biotechnology company that lists government “cost containment” efforts as a risk to its bottom line.
Another committee member reported consulting income from 11 to 13 pharmaceutical companies, including eight that Medicare records show have earned millions billing for drug waste. His pharma ties were disclosed in in 2019 through this year.
Those committee members said they reported relevant relationships to the national academies and that the information is readily available outside of the report.
What’s more: The National Academy of Sciences itself for years has been collecting generous gifts from foundations, universities and corporations, including at least $10 million from major drugmakers since 2015, its treasurer show. Among the donors are companies with millions to retain or lose over the drug waste committee’s findings.
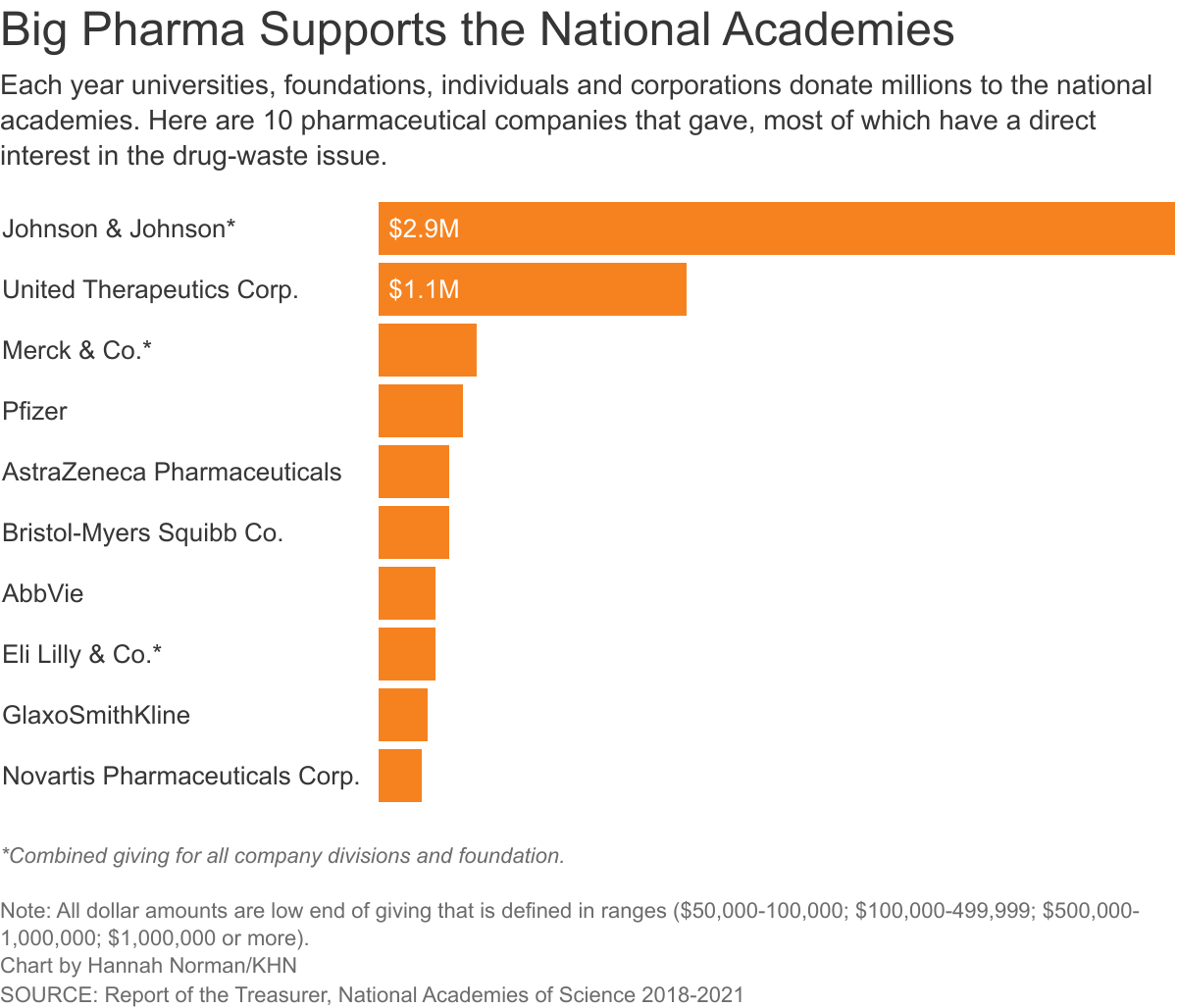
The fact that those relationships were not disclosed in the final report by an organization charted in 1863 to advise the nation amounts to “egregious” failures, said Sheldon Krimsky, a Tufts University professor and expert on conflicts of interest in science.
“The amount of money you’re reporting is really substantial,” he said. “It really raises questions about the independence” of the national academies.
In a statement emailed to KHN, the national academies said the two members with undisclosed board and consulting roles had “no current conflicts of interest during the time the [drug waste] study was being conducted” from January 2020 through February. The report did for two others on the 14-member board. The report in question was paid for by federal officials, and “funds from for-profit organizations with a direct financial interest in the outcome of a study may not be used to fund advisory consensus studies, except in rare circumstances,” national academies spokesperson Dana Korsen said in the emailed statement.
She also said the organization is implementing a new conflict-of-interest policy that will be fully in place this fall.
“Protecting the integrity, independence, and objectivity of our study process is of the utmost importance to the National Academies,” her statement said.
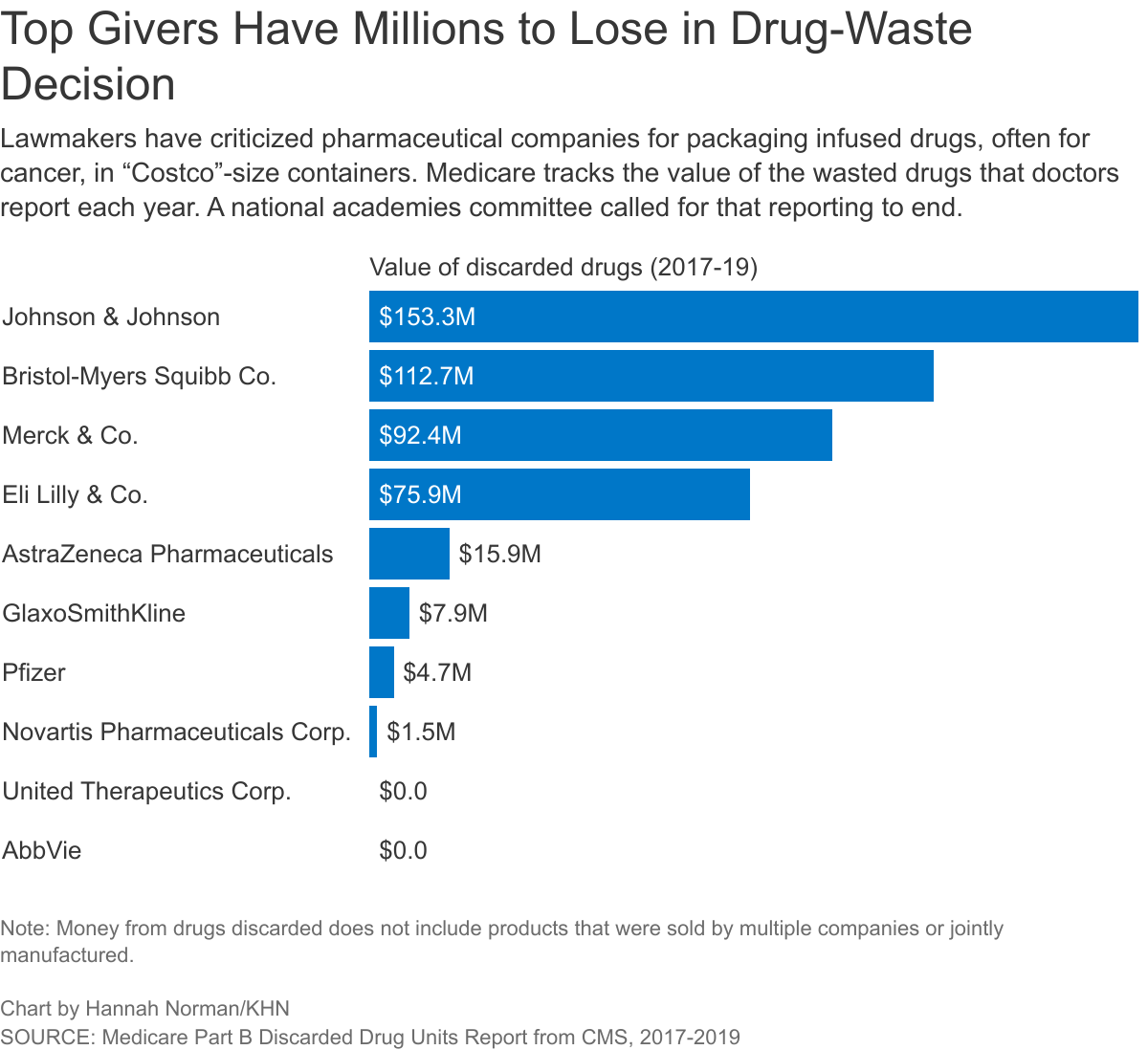
The committee’s failure to call for concrete changes — and the millions in gifts from pharmaceutical companies to the national academies — looked familiar to David Mitchell, president of Patients for Affordable Drugs and a cancer patient who relies for his survival on a drug with high waste costs.
“We have found in our work that pharma is like an octopus,” he said, “and at the end of each tentacle is a wad of cash.”
Waste Shocked Policymakers in 2016
Dr. Peter Bach and colleagues published an explosive in 2016 that for the first time showed that taxpayers and health insurance rate payers were bankrolling an estimated $2.8 billion a year in drug waste. The findings encompassed all U.S. health care — not just what’s reported by doctor’s offices to Medicare — and were covered in the .
Bach, a researcher with the Center for Health Policy and Outcomes at Memorial Sloan Kettering Cancer Center, found that medications infused in doctors’ offices often arrived in vial sizes fit for a linebacker but might be given to a waif. Given sterility and other concerns, the extra milligrams, often for cancer therapies that can cost thousands of dollars per dose, were typically discarded.
Congress and policymakers took notice.
In 2017, Sens. Amy Klobuchar (D-Minn.) and Chuck Grassley (R-Iowa) urging health care agencies to develop a “joint action plan” to address the waste. Sens. Dick Durbin (D-Ill.) and Rob Portman (R-Ohio) introduced an even stronger measure in and again that would allow Medicare to recoup the cost of the wasted drugs. None of the bills has passed.
The refund mandate made it into a broader drug pricing measure that also failed, but not before the Congressional Budget Office took a in 2020 and estimated $9 billion could be saved over a decade.
Medicare officials also urged doctors to use a billing code to document the amount taxpayers were spending on wasted drugs each year — which amounted to $753 million in 2019 alone, shows.
Before and while Bach’s paper was making waves, physicians who would eventually be on the national academies committee were forging alliances with the pharmaceutical industry.
Dr. Kavita Patel reported earning a speaking fee in 2015 from the Pharmaceutical Research and Manufacturers of America, or PhRMA, of $5,001 to $15,000. She also accrued assets valued at more than $50,000 for her role as a pharmaceutical company board member, according to 2015 and 2018 disclosures filed with the Government Accountability Office.

Dr. Anupam Jena, who also served on the committee, wrote a 2018 article with staff members of PhRMA that medications should be valued not for their actual benefit, but rather for the potential for innovation that comes with making new therapies.
The ‘Kiss of Death’
In 2016, lawmakers an independent study of the drug waste. In September 2019, the National Academy of Sciences $1.2 million to complete the report.
At the outset of its study in January 2020, national academies committee members declared their potential conflicts of interest in a closed session, according to the meeting agenda.
Bach was among the physicians and other experts who later presented to the national academies committee. He said his team had laid out two possible solutions from the start: Have companies make a variety of vial sizes to minimize waste, or pursue refunds.
Former Medicare administrator Donald Berwickpresented to the committee at a June 2020, virtual meeting, exhorting its members to defy the expectation that they’d be one more committee that failed to do anything meaningful about health costs.
“Someone’s got to begin to set a standard and say, ‘Nope, this money is too important for … us to accede to this,’”
The report’s recommendations were “the result of extensive fact-finding, full committee discussions and unanimous consensus,” said committee chairperson Dr. Edward Shortliffe, chair emeritus and adjunct professor in the Department of Biomedical Informatics at Columbia University.
The report, though, did not meet Berwick’s call to action. In a webinar summarizing the report findings, Jena as valuable enough to justify the total cost of each vial, completely used or not. Patel and others summarized the findings in a , saying the committee argued against tracking the money wasted and instead called for a “” approach.
Bach said the conclusions were “better than pharma could have ever hoped for” and called the whole-of-government idea the “kiss of death.”
Berwick said that he was “disappointed” by the conclusions and that all committee members’ industry relationships should have been reported. He noted that, in his experience, committee members have been very open about conflicts and the national academies dismissed those who had them.
Presented with KHN’s findings about certain committee members’ undisclosed pharmaceutical company income and consulting relationships, Bach said they raise serious concerns.
“The conflicts align just way too closely with the results,” he said. “That’s what makes it hard to ignore.”
‘Current’ Conflicts Don’t Tell Full Story
Conflicts of interest became a hot topic more than a decade ago, amid a series of over Big Pharma quietly backing influential doctors.
Reforms followed, with countless medical journals, nonprofits and government agencies strengthening their conflict-of-interest policies.
The national academies came and 2016 for failing to disclose conflicts among committee members advising federal officials and in 2017 on genetically modified crops.
Its webpage on conflicts underscores why strong disclosure rules are important: “The institution should not be placed in a situation where others could reasonably question, and perhaps discount or dismiss, the work of the committee simply because of the existence of such conflicting interests.”
Yet conflict-of-interest experts interviewed by KHN said the national academies stands out by considering only “current” conflicts and not those going back three years, as is more typical. Korsen said the National Academy of Sciences is working toward requiring five years of disclosures.
Several experts said that, given the trust placed in — and $200 million in federal funding awarded to — the national academies, a number of conflicts should have been disclosed in the report.
They include those of Patel, who is described in her report biography as a Brookings Institution fellow, a primary care physician in Washington, D.C., and former Obama administration policy adviser.
The national academies declined to provide the conflict-of-interest form that Patel or any other member filled out at the outset of the committee’s work in early 2020.
Unrelated Securities and Exchange Commission records show that, before she joined the committee in 2020, Patel’s role as a for Tesaro, a developer of cancer medications, became very lucrative when GlaxoSmithKline bought the company. At the time of the 2019 sale, Patel was in line to receive an estimated $1.4 million for her shares and stock options, according to a December 2018 Tesaro .
Also in 2020, Patel was appointed to the board of Sigilon Therapeutics, a biotech company with no product on the market. The company awarded her stock options then worth an estimated $369,000, an SEC .
Sigilon described state and federal efforts to control costs as a risk to its business in an annual : “Any cost containment measures could significantly decrease … the price we might establish for our products.”
The national academies’ lack of disclosure of those roles “to me is a violation of almost all the standards that I’m aware of for disclosing conflicts of interest,” said Krimsky, of Tufts.
Patel told KHN she “fully and transparently participated” in the disclosure process and “provided all of the information requested.” She said: “In addition, many of the financial relationships incurred over the course of my work had already been disclosed in the public record.”
Patel was the lead writer on the Feb. 25 opinion that summarizes the committee’s report as focusing on the need to reduce inefficiencies, “rather than on trying to recover from pharmaceutical companies the financial worth of the portion of drug that was not used.”
Patel said she was “objective in all of my contributions” to the national academies report.
The national academies — as an organization — reported in its 2016 treasurer report that while 84% of its funding in 2011 was from federal agencies, the amount was failing. So it was working to “grow the non-federally sponsored work.”
“It will be very important for the future of the institution to continue vigorous efforts to diversify its sources of income,” the says.
A KHN review of treasurer reports from 2015 through 2020 shows that pharmaceutical companies have given consistently to the national academies. Drugmakers donated at least $10 million over those years. Their giving is reported in ranges, often $100,000 to $500,000, and that total assumes they gave the lowest amount in each range each year.
A 2018 treasurer recognized Merck & Co. for more than $5 million in cumulative giving and 10 other drugmakers for donating more than $1 million.
None of those donations was listed in the drug waste report. But listing them would reassure readers, said Genevieve Kanter, a University of Pennsylvania assistant professor of medical ethics and health policy.
“If the national academies is interested in producing a credible, independent report,” she said, “I think they would report all of those donations in the report itself.”
Jena, a Harvard Medical School associate professor, physician at Massachusetts General Hospital and an economist, also had no conflicts disclosed in the report.
Jena has disclosed consulting a dozen major pharmaceutical companies, articles in the Journal of the American Medical Association and The BMJ show. Most of those companies have a direct financial interest in the drug waste matter, a KHN review of Medicare data shows. He said he disclosed all his consulting relationships to the national academies.
After the report came out, he took the lead on a Health Affairs that says Medicare should stop tallying the waste money.
“Attempts by public payers to recoup overpayments are unlikely to be successful since they may simply end up paying higher prices” if drugmakers raise the price tag for the medications.
That article initially omitted his consulting relationships with numerous pharmaceutical companies — but journal editors updated the disclosures after KHN inquired.
Jay Hancock and Megan Kalata contributed to this report.
This <a target="_blank" href="/news/national-academies-big-pharma-support-drug-waste-report/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1350718&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>He sent cards in the mail advertising air purifiers using ActivePure technology to new homeowners: “KILL COVID-19, CORONAVIRUS IN YOUR HOME!!”
One card landed on the desk of a postal inspector, who called it false and misleading in a court record. Shumaker then told an undercover agent on the phone on April 24, 2020, that the air purifier “kills the Coronavirus Virus on the spot,” according to a .
Weeks later, as Shumaker was heading out the door to his daughter’s tennis tournament, eight law enforcement officers detained him. In August, he to distributing “a pesticide device that was misbranded in that the product label was missing an EPA establishment number.” In other words, he failed to follow the letter of a little-known law.
Shumaker told KHN he was just a salesperson and the devices were being shipped straight from the manufacturer. “So I don’t know т what am I supposed to do?” he asked. “How do I know if there’s a sticker on there or not? I don’t have a clue.”
The company that makes the devices, ActivePure Technologies, said Shumaker was not an authorized or known salesperson of its products.
The sting is a rare example of enforcement in an arena where money is gushing like a geyser but oversight is nearly nonexistent. Electronic air cleaners, heavily marketed to gyms, doctors’ offices and hospitals, companies and schools awash in federal covid relief funds, tend to use high-voltage charges to alter molecules in the air. The companies selling the devices say they can destroy pathogens and clean the air.
But academic air quality experts say the technology can be ineffective or potentially create harmful byproducts. Companies that make the devices are subject to virtually no standardized testing or evaluation of their marketing claims. A KHN investigation this spring found that over 2,000 schools across the country have bought such technology.
“That’s one of the reasons these companies thrive, is that there’s nobody, nobody checking every aspect of what they do,” said , a Colorado State University associate professor who specializes in atmospheric and indoor chemistry.
Regulatory Patchwork
An alphabet soup of federal agencies have truth-in-advertising or product medical device oversight powers but have done little about air cleaners or left broad loopholes. That has left a handful of states to take the most decisive action on the industry.
The Centers for Disease Control and Prevention does not regulate the devices but, like academic air quality experts, time-tested portable HEPA filters to clean the air in rooms. In comparison, ionizing and dry hydrogen peroxide air purifiers have a “less-documented track record” in air cleaning, the CDC says.
The CDC also urges consumers to research the technology and “request testing data.” Those reports, though, can be difficult to parse. They include arcane terms like “natural decay” and test conditions that only an expert could spot as different from those that prevail in real life.
The Food and Drug Administration regulates medical devices. But only air purifiers for a direct medical use or that make a medical claim, like relieving allergies, qualify. The FDA doesn’t consider ads saying a device can kill a microorganism a “medical claim,” spokesperson Shirley Simson said in an email.
Instead, the air purifiers fall under the Environmental Protection Agency’s authority as marketed to destroy “pests,” which include bacteria or viruses. But “unlike chemical pesticides, the EPA does not register devices and, therefore, does not routinely review their safety or efficacy,” said.
Trying to fill the gaps, California bans the sale of air purifiers than a certain level of the toxic ozone gas. The New York State Education Department is “” that schools buy air purifiers it describes as “ion generators” or “corona discharge technology.”
, a University of Toronto civil engineering professor who studies indoor air quality, said more meaningful national regulation might clarify for consumers how the devices would work in an actual room.
“If you get any serious government oversight, a big chunk of this industry will go away,” said Siegel.
тI Was Alone’
While “pesticide” might evoke the idea of a roach killer, the EPA applies the term more broadly: A pesticide is any substance that claims to kill or mitigate pests. Technologies that claim to do the same through physical means т including air purifiers that inactivate bacteria and viruses т are considered .
And while the agency requires proof that pesticides in its premarket review, it has no such requirement for so-called pesticide devices т such as electronic air cleaners that deploy ions or “reactive oxygen” to purify the air.
Instead, manufacturers need to obtain what’s known as an indicating where the device is made, and then they and their sellers must label their products with it. That’s the step Shumaker pleaded guilty to skipping.
“There is no review associated with that,” said , a lawyer who specializes in chemical regulation law at Keller and Heckman in Washington, D.C. “That is automatic. It’s like trying to get license plates for your car.”
So Shumaker told KHN he was baffled as to why he was targeted instead of the corporate level, which in this case would be the company, Aerus, which Technologies. Dr. Deborah Birx, former adviser to President Donald Trump, joined ActivePure in March as chief medical and science adviser.
“I was alone,” Shumaker said in an interview about facing charges. “Nobody backed me up.”
Joe Urso, CEO of ActivePure Technologies, told KHN that its studies showing its devices inactivate the virus that causes covid were not completed until the fall, long after the postcards were sent. Urso said in a statement that his company’s devices do have establishment numbers, and that he supports the ruling against Shumaker.
Federal Trade Commission officials have written warning letters companies during the pandemic. The commission requires claims about a product’s safety and efficacy to be supported with “competent and reliable scientific evidence.”
One of the last high-profile actions the FTC took against an air purifier company was in 1997, when the Justice Department on its behalf against Alpine Industries, which made ozone-generating air purifiers. In 2001, a judge fined Alpine $1.49 million for failing to stop making unsubstantiated claims about its devices, which it had said relieved allergies and removed indoor pollutants.
Alpine is a related company to , according to the FTC. And a majority of EcoQuest International assets were bought in 2009 , according to its 2021 press kit. ActivePure makes the device Shumaker got into trouble for selling.
Siegel, of the University of Toronto, consulted with U.S. government agencies targeting the misleading marketing claims of some air-cleaner companies about 10 years ago. He finds the company-by-company approach to be a game of “whack-a-mole.”
“A company goes away because they have regulatory scrutiny and reinvent themselves a few months later,” he said. “The only solution I see to this problem is a government agency really takes ownership of this т the information dissemination to consumers and the claims by manufacturers. I see no other path forward.”
тIt’s Just Buyer Beware’
The Federal Insecticide, Fungicide, and Rodenticide Act, which regulates pesticide devices, was written decades ago and applied to things such as flypaper, long before anyone anticipated machines that would blast ions to clean the air.
“We’re just pushing EPA to try and get updated,” said Patrick Jones, president of the . “It’s just buyer beware.”
Even before the covid pandemic, Jones’ group was ringing the alarm over the increasing public health claims around pesticide devices. The pesticide control association wrote in a 2019 to the EPA of its concern about the growing use in health care facilities of “non-government evaluated pesticide devices that make unsubstantiated human health claims тІ with no scientific data being submitted to EPA to prove their effectiveness.”
EPA spokesperson Tim Carroll said in an email that the agency is developing more outreach materials for schools on air purifiers.
But as few independent authorities assess the effectiveness of the devices, school officials have been snapping them up.
Last summer, the private St. Thomas More School in Kansas City, Missouri, bought ionizing air purifiers to fight covid. Scott Dulle, the director of building and grounds, said he went with technology he saw health leaders buying.
“We followed the doctors and the hospitals and the government,” he said. “They would not put their patients and staff in harm’s way.”
AAPCO’s Jones said changes to federal oversight are needed to better deal with the flood of devices. His solution: If a pesticide device makes a public health claim, it should be evaluated with the same rigor used for pesticides like ant spray.
But to alter the law fundamentally would take congressional action, EPA’s Carroll said.
The EPA pesticide device companies and sellers if a product makes misleading or false claims т and fines can reach into the millions, according to Brandon Neuschafer, a lawyer who specializes in agricultural regulations at the St. Louis-based firm Bryan Cave Leighton Paisner. He noted companies are often turned in by their competitors.
Last fiscal year, Carroll said, the agency issued 19 import refusal notices and sent six advisory letters for covid-related air-purifying products т a small fraction of its 2020 pesticide actions. Carroll said such investigations are ongoing and a high priority.
But EPA’s resources were not the same as they were many years ago, Neuschafer said, as the agency is working with smaller staffing and budgets.
тWorse Indoor Air Quality’
Almost a decade before covid emerged, New York’s education department asked state health officials an AtmosAir bipolar ionization unit to see if it would improve the air quality.
During a test in an empty classroom, they found that levels of harmful ozone gas and “ultrafine particles” that can cause were elevated, indicating “worse indoor air quality when the AtmosAir Bipolar ionization unit is operating,” the 2013 state Bureau of Toxic Substance Assessment .
New York State Department of Health officials released the study in response to a KHN public records request about the education department’s covid-era guidance, which urges schools .
AtmosAir spokesperson Sarah Berman said the device studied in 2013 is discontinued and “all current products have no affiliation to” it. She also said in an email that tests by third-party labs found that “our bipolar ionization products do not contribute to unacceptable levels” of volatile organic compounds, which are potentially harmful substances.
The California Department of Public Health advised in September “air cleaning devices that generate harmful pollutants (i.e., ionization devices or ozone generators)” on the third page of a single-spaced, 44-page document. That guidance was widely overlooked. Districts from to to bought ionization systems.
But the state does have a one-of-a-kind law: It bans air purifiers that emit anything above a certain level of ozone.
New Jersey doesn’t have the same kind of regulation: that a public school district there bought of ozone-emitting Odorox devices on the California Air Resources Board’s list of “” air purifiers. Since then, the New Jersey health department warning schools about the air purifiers “that may harm health,” listing the specific hazards of ozone to children’s health.
Back in Georgia, Shumaker more than $9,000 and is on two years’ probation.
And the postcards that got him into trouble? Those led to only a handful of sales.
“So it was just like setting money on fire,” he said.
This <a target="_blank" href="/public-health/government-oversight-of-covid-air-cleaners-leaves-gaping-holes/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1338711&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>The new standard would require employers to remove workers who have covid-19 from the workplace, notify workers of covid exposure at work and strengthen requirements for employers to report worker deaths or hospitalizations to the Occupational Safety and Health Administration.
“These are the workers who continue to go into work day in and day out to take care of us, to save our lives,” said Jim Frederick, acting assistant secretary of Labor for occupational safety and health. “And we must make sure we do everything in our power to return the favor to protect them.”
set to take effect immediately after publication in the Federal Register and are expected to affect about 10.3 million health care workers nationwide.
The government’s statement of reasons for the new the work of KHN and The Guardian in tallying health care worker covid deaths . Journalists documented far more deaths than the limited count by the Centers for Disease Control and Prevention, which through May tallied 1,611 deaths on case-reporting forms that were often incomplete.
The Lost on the Frontline project documented early calls for better respiratory protection for health care workers than loose-fitting face masks, noted serious complaints to OSHA from hospital workers that went unaddressed and revealed repeated employer failures to report dozens of worker deaths. It also found that health care employers were often remiss in notifying workers about exposure to the coronavirus on the job.
The new standard would address some of those problems.
The rules require workers to wear N95 or elastomeric respirators when in contact with people with either suspected or confirmed covid. They strengthen employer record-keeping requirements, saying employers must document all worker covid cases (regardless of whether they were deemed work-related) and report work-related deaths even if they occur more than 30 days after exposure.
Until now, employers were required to report a hospitalization only if it came within 24 hours of a workplace exposure. Now all work-related covid hospitalizations must be reported. The rules also mandate notification about exposure to a sick colleague, patient or customer if the worker was not wearing a respirator.
There is a lot to like about the new rule т except for the timing, according to Barbara Rosen, vice president of the Health Professionals and Allied Employees union in New Jersey.
“It’s a little late,” she said. “If we had had this in place at the beginning, it would have saved a lot of lives and a lot of suffering that has gone on with health care workers and probably patients in hospitals because of the spread.”
She said she was pleased with the requirement that workers be paid when they isolate with covid and that employers formulate a detailed covid plan with the input of non-managers.
The day after he took office, President Joe Biden issued an calling on OSHA to “take swift action to reduce the risk that workers may contract COVID-19 in the workplace.” The rule has been for coming late т about which Labor Department officials said on a press call that such standards typically take years, not months, to formulate. It has also been derided for failing to enact requirements on employers outside of health care.
“OSHA’s failure to issue a COVID-specific standard in other high-risk industries, like meat and poultry processing, corrections, homeless shelters and retail establishments is disappointing,” according to a statement from David Michaels, a former OSHA administrator and professor with the George Washington University School of Public Health. “If exposure is not controlled in these workplaces, they will continue to be important drivers of infections.”
The new rule also cites 67,000 worker complaints during the pandemic, with “more complaints about healthcare settings than any other industry.” The rule would protect workers from retaliation for staying home when sick with covid, alerting their employer about a covid hazard or exercising their rights under the emergency rule.
Through March 7, about half of health care workers said they had received at least their first dose of a covid vaccine, according to a . About one-third of those polled said they were unsure if they would get a vaccine. The issue has been controversial, especially in Houston, where workers staged a protest over their employer’s vaccine mandate.
The new rules exempt some office-based health care workplaces where all staff members are vaccinated and measures are taken to screen people with potential illness. The rule summary estimates the measures will prevent 776 deaths and 295,000 infections.
The new rule also will “enable OSHA to issue more meaningful penalties for willful or egregious violations, thus facilitating better enforcement and more effective deterrence against employers who intentionally disregard тІ employee safety.”
Kristin Carbone said the measure came too late for her mother, Barbara Birchenough, 65, a New Jersey hospital nurse who’d asked family members to gather gardening gloves and trash bags to serve as makeshift personal protective equipment before she fell ill and later died on April 15, 2020. Still, she said, it’s a necessary step.
“If there is a silver lining,” she said, “I’m glad that out of this tragedy come positives for the people that are left behind.”
бюЙѓхњДЋУНвюl Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFFтan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/news/labor-department-osha-emergency-rules-protect-health-care-workers-from-covid/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1323957&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>Dr. Janet Woodcock, acting commissioner of the Food and Drug Administration, told state officials during a White House call Tuesday that they could store expired doses until new data shows whether the vaccines are safe to use, according to multiple state officials.
State health officials have vaccine administrators against using expired doses. Now, though, the FDA appears optimistic that the Johnson & Johnson expiration dates т which begin to kick in later this month т could be extended, according to state officials who were on the call.
“This is really welcome news,” said Dr. Joseph Kanter, state health officer for the Louisiana Department of Health. Louisiana has 14,000 J&J doses that will expire this month. “I think at the end of the day there’ll be less waste.”
The federal government has delivered 21.4 million doses of the company’s vaccine to states, but just more than half т 11.2 million т have been administered, the Centers for Disease Control and Prevention. The quantity is a fraction of shipments of Pfizer-BioNTech’s covid vaccine, which are approaching 200 million doses, as well as Moderna’s shot, which stands at more than 150 million doses.
As demand for vaccination has dwindled across the nation, state officials have stepped up public pleas for holdouts to get a shot. They’ve held discussions with the Biden administration about how to avert a glut of J&J doses т hundreds of thousands at a minimum т from going to waste. On Monday, Ohio Gov. Mike DeWine said 200,000 J&J doses would expire June 23 and the state had no legal way to send unused doses to other states or countries.
Through the Trump administration’s Operation Warp Speed initiative, the federal government awarded J&J a $1 billion to deliver 100 million doses of its covid vaccine.
The J&J single-dose vaccine lasts three months under refrigeration and two years frozen. Extending the expiration date is seen as a more feasible option for quickly preserving thousands of doses, as opposed to redistributing them to other states or countries, state officials say.
“There aren’t that many states right now that are needing more vaccine than what they have in hand,” said Dr. Marcus Plescia, chief medical officer of the Association of State and Territorial Health Officials. “There’s enough out there.”
Federal officials believe data about the expiring June doses from an ongoing stability study will come in in roughly a month, two state officials said.
“We also continue to conduct stability testing with the goal of extending the amount of time our covid-19 vaccine can be stored before expiry,” a J&J spokesperson said. “We will share further information as we are able to.”
The White House declined to specify the number of J&J doses nationally that will expire this month. The FDA declined to comment on Woodcock’s remarks.
Once viewed as crucial to the U.S. vaccination effort for persuading on-the-fence people to get the single-shot dose, J&J has played a modest role. Officials partly attribute that to federal regulators temporarily halting its use in April after reports of rare but serious blood clots.
“That just appears to have slowed demand,” Plescia said. The company previously said it would deliver the 100 million doses by the end of June.
As far as Jim Mangia, chief executive of St. John’s Well Child and Family Center, can tell, the demand ground to a halt once the FDA OK’d resuming use of the J&J vaccine. Mangia said his network of 26 clinics in the Los Angeles area has more than 14,000 doses on hand that county officials have been unwilling to take back.
He said patients who liked the one-shot benefit requested it before the safety concerns, but since then there have been no requests. “Whenever we offer it, everyone says no,” he said.
Mangia said his clinic network is seeing overall distribution of the vaccines remain steady as sites expand hours and offer Friday night vaccine events for those concerned about missing work because of possible side effects. But given the lack of interest in J&J’s vaccine, he said, he doesn’t think moving the expiration date will improve matters.
Officials in West Virginia have more than 20,000 doses of J&J’s vaccine on hand but little backlog of other covid vaccines, said state covid czar Dr. Clay Marsh, who is also vice president of West Virginia University Health Sciences. Officials offered the excess to other states, but there were no takers. Marsh said they approached the federal government about sending the unused doses to countries that need it, but have learned the logistics are challenging.
“If we’re not able to use something that can save lives, we’re trying to see if there’s someone who can,” he said.
As of Tuesday, 52% of Americans had received at least one dose of covid vaccine, according to the CDC.
Officials have also engaged in public finger-pointing about the expiring doses, with some state and local officials calling for more federal help to redistribute the doses already delivered. Meanwhile, Andy Slavitt, White House senior adviser for the covid-19 response, noted in a Tuesday call with reporters that vaccine doses ordered by state officials “should end up in people’s arms” and governors should work directly with the FDA on proper storage.
“There are plenty of people across the country, in every state, that still haven’t been vaccinated, that are eligible, that are at risk and need to get vaccinated,” he said.
бюЙѓхњДЋУНвюl Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFFтan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/public-health/unused-johnson-johnson-covid-vaccine-doses-expiration-backlog/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1322274&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>Boeing noted that “air ionization has not shown significant disinfection effectiveness.”
Companies that make the air purifiers say they emit charged ions, or “activated oxygen,” that are said to inactivate bacteria and viruses in the air. Boeing did not test the technology’s effectiveness in the air, only on surfaces. It also used a “surrogate” for the virus that causes covid-19.
has been cited in a federal lawsuit filed by a Maryland consumer against Global Plasma Solutions, maker of the “needlepoint bipolar ionization” technology that a Boeing spokesperson said its engineers tested.
The proposed class-action lawsuit says GPS makes “deceptive, misleading, and false” claims about its products based on company-funded studies that are “not applicable to real world conditions.”
A GPS spokesperson said the lawsuit is “baseless and misleading” and that the company will aggressively defend against it. He added that Boeing “researchers deemed the study тinconclusive.’”
“Plaintiff’s Complaint throws the proverbial kitchen sink at GPS in the hopes that something might stick,” the air purifier company says in filed May 24 as part of its motion to dismiss the proposed class action. “But it is devoid of any concrete, specific allegations plausibly alleging that GPS made even a single false or deceptive statement about its products.”
The plaintiff’s case cites that found that more than 2,000 U.S. schools had bought air-purifying technology, including ionizers. Many schools used federal funds to purchase the products. In April, a covid-19 commission task force from The Lancet, a leading medical journal, composed of top international health, education and air-quality experts, called various air-cleaning technologies т ionization, plasma and dry hydrogen peroxide т “.”
Boeing said in that with ionization there is “very little external peer reviewed research in comparison to other traditional disinfection technologies” such as chemical, UV and thermal disinfection and , all of which it relies on to sanitize its planes.
The controversy is getting the attention of school officials from coast to coast. They include one California superintendent who cited the lawsuit and switched off that district’s more than 400 GPS devices.
For worried parents and academic air-quality experts who regard industry-backed studies with skepticism, the Boeing report heightens their concerns.
“This [study] is totally damning,” said Delphine Farmer, a Colorado State University associate professor who specializes in atmospheric and indoor chemistry who reviewed the Boeing report. “It should just raise flags for absolutely everyone.”
тNo Reduction’ in Bacteria
GPS pointed to another study, one conducted in the weeks before Boeing began its study in September, by a third-party lab. It completed of т powered by GPS technology т that another aviation company now markets to clean the air and surfaces in planes.
That study looked at the effect of the ionizers on the virus that causes covid-19 when used on aluminum, a type of plastic called Kydex and leather. The test it was conducted in a sealed, 20-by-8-foot chamber, with airflow speeds of 2,133 feet per minute т or about 24 mph. At the end of 30 minutes, “the overall average decrease in active virus” was more than 99%.
“Given the specific environment this was tested in, the quality of the materials, and the method in which the virus was dispersed, it is safe to say that the bipolar ionization system used in this experiment has the ability to deactivate SARS-CoV-2 with the given ion counts,” the Aug. 7 report from the third-party lab says.
The following month, Boeing of GPS devices and another kind of ionization technology.
The Boeing study cites a GPS that says its device killed 99.68% of E. coli bacteria in one test in 15 minutes. GPS records show the test was done on bacteria suspended . The Boeing engineers used the company’s technology to try to kill E. coli on surfaces in a lab but found “no observable reduction in viability” after an hour.
The Boeing study notes it “was unable to replicate supplier results in terms of antimicrobial effectiveness.”
GPS cautioned that the Boeing tests examined disinfection of surfaces, not the air: “While GPS products do have the ability to help reduce pathogens in air and on surfaces, GPS products are not chemical surface disinfectants.”
Yet surface tests comprise half of the test results the company lists on its “” webpage, a GPS spokesperson confirmed.
Boeing researchers found another lab result they could not replicate: While the GPS white paper a 96.24% reduction in Staphylococcus aureus in 30 minutes, Boeing engineers found “no reductions” in the bacteria in an hourlong test.
Boeing found minimal or no reduction on surfaces in four other pathogens it tested with GPS ionizers for an hour in a Huntsville, Alabama, lab.
Notably, Boeing’s tests in Huntsville detected no hazardous ozone gas from the GPS unit, the . The “corona discharge” ionization technology from another vendor that Boeing also studied did emit ozone at levels that “exceeded regulatory standards.”
A University of Arizona lab test described in the Boeing study found that the GPS device showed a 66.7% inactivation of a common cold coronavirus on a surface after an hour of exposure at up to 62,000 negative ions per cubic centimeter. That ion level is far higher than the amount of ions company leaders have said the devices tend to deliver to a typical room. Those levels have ranged from to and 30,000 ions per cubic centimeter when an HVAC system is running, according to records and statements made by .
In during a Berkeley Unified School District meeting in California, a physicist with executives said a level of more than 60,000 ions per cubic centimeter “has been shown to be not healthy.”
GPS noted that Boeing deemed the 66.7% effectiveness rate in killing the common cold virus “statistically significant.” A GPS spokesperson said the result validates needlepoint bipolar ionization’s “effectiveness against certain pathogens.” In its report, Boeing called the test results “inconclusive” due to “lack of experimental confirmation.”
A GPS spokesperson also highlighted a passage in the Boeing report’s conclusion that said: “There remains significant interest in air ionization due to lack of byproduct production, minimal risk to human health, minimum risk to airplane materials and systems, and the potential for persistent disinfection of air and surfaces under specific flow conditions.”
The Boeing study concluded in January. In April, GPS of additional tests it funded at a third-party lab showing its technology “is highly effective in neutralizing the SARS-CoV-2 pathogen.”
Boeing engineers said their study highlights the need for those in the ionization business to standardize the evaluation of the technology “to allow comparison to other proven methods of disinfection.”
Ripple Effects of the Boeing Study
On May 7, law firms representing a man who spent over $750 on a GPS air cleaner in Texas filed the “fraudulent concealment” in U.S. District Court in Delaware.
The lawsuit claims that the defendant’s “misrepresentations and false statements were woven into an extensive and long-term advertising campaign … accelerating during the COVID-19 pandemic.”
“People are being victimized by these companies for profit,” said Mickey Mills, a Houston attorney for the plaintiff. “People are scared because of covid, and they capitalize on it.”
In filing a motion to dismiss the case, GPS told the court the lawsuit was an “attempt to distort the facts and assert baseless claims, doing grave damage to GPS’s business in the process.”
The GPS court document also says the disclaimers on its website “make it unreasonable for any consumers to believe that the efficacy demonstrated in GPS studies will necessarily be the same for their particular application.”
It asserts that most of the GPS statements identified in the plaintiff’s lawsuit т such as “safe to use” and “cleaner air” т amount to “non-actionable puffery” as they are “vague generalities and statements of opinion.”
The lawsuit spurred a Newark, California, school district to turn off its GPS devices, according to a from Superintendent Mark Triplett to district families. The district spent nearly $360,000 on the devices, an April shows.
The roughly 5,500-student district bought GPS units for every school HVAC system, Triplett said in a March school in which he noted the technology “arguably is much better than any filter.” By May, in the memo the district had become aware of the lawsuit “alleging the misrepresentation” of the devices and would continue to monitor the situation.
A company spokesperson noted GPS appreciates Newark’s concerns and has reached out to share additional data and answer questions, as well as extended “an offer to conduct onsite testing to verify the safety of this technology and the added benefits.”
Megan McMillen, vice president of the Newark Teachers Association and a special education preschool teacher, said it was disheartening to know the cash-strapped district in the Bay Area spent so much on the devices instead of other safety measures or services to mitigate learning loss after the chaotic pandemic year.
“For such a big chunk of that [money] going to something potentially ineffective … is really frustrating,” she said.
бюЙѓхњДЋУНвюl Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFFтan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/public-health/boeing-tested-air-purifiers-like-those-widely-used-in-schools-it-decided-not-to-use-them-in-planes/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1311951&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>[АфДЧАљАљБ№ГІГйОБДЧВд:Ь§This article was revisedТ at 4:15 p.m. ET on May 7, 2021, to reflect that an Odorox distributor,Т not Pyure, commissioned the ozone test on a Boss XL3.Т And the article was revised to reflect thatТ PyureТ CEO Jean-Francois “JF” Huc said his company provides stringent operating guidelines for use of the company’s air purifiers but did notТ acknowledge thatТ school staffers are often not warned about the problems they could face if a too-powerful device is used in a too-small room.]
Last summer, Global Plasma Solutions wanted to test whether the company’s air-purifying devices could kill covid-19 virus particles but could find only a lab using a chamber the size of a shoebox for its trials. In the , the virus was blasted with 27,000 ions per cubic centimeter.
In September, the company’s founder that the devices being offered for sale actually deliver a lot less ion power т 13 times less т into a full-sized room.
The company nonetheless used т over 99% viral reduction т in marketing its device heavily to schools as something that could combat covid in classrooms far, far larger than a shoebox.
School officials desperate to calm worried parents bought these devices and others with a flood of federal funds, installing them in more than 2,000 schools across 44 states, a KHN investigation found. They use the same technology т ionization, plasma and dry hydrogen peroxide т that Commission recently deemed “often unproven” and potential sources of pollution themselves.
In the frenzy, schools are buying technology that academic air-quality experts warn can lull them into a false sense of security or even potentially harm kids. And schools often overlook the fact that their trusted contractors т typically engineering, HVAC or consulting firms т stand to earn big money from the deals, KHN found.
Academic experts are encouraging schools to pump in more fresh air and use tried-and-true filters, like HEPA, to capture the virus. Yet every ion- or hydroxyl-blasting air purifier sale strengthens a firm’s next pitch: The device is doing a great job in the neighboring town.
“It’s a self-fulfilling prophecy. The more people buy these technologies, the more they get legitimacy,” said, a civil engineering professor at the University of Toronto. “It’s really the complete wild west out there.”
Marwa Zaatari, a member of the American Society of Heating, Refrigerating and Air-Conditioning Engineers’ (ASHRAE) Epidemic Task Force, first compiled a list of schools and districts using such devices.
Schools have been “bombarded with persistent salespersons peddling the latest air and cleaning technologies, including those with minimal evidence to-date supporting safety and efficacy” according to Thursday by the Center for Green Schools and ASHRAE.
Zaatari said she was particularly concerned that officials in New Jersey thousands of devices made by another company that emit ozone, which can exacerbate asthma and harm developing lungs, according to decades of research.
“We’re going to live in a world where the air quality in schools is worse after the pandemic, after all of this money,” Zaatari said. “It’s really sickening.”
The sales race is fueled by roughly $193 billion in federal funds allocated to schools for teacher pay and safety upgrades т a giant fund that can be used to buy air cleaners. And Democrats are pushing for $100 billion more that could also be spent on air cleaners.
In April, Global Plasma Solutions said further tests show its covid in the air and on surfaces in larger chambers. The company studies still use about of ions than its leaders the devices can deliver, KHN found.
There is virtually no federal oversight or enforcement of safe air-cleaning technology. Only California bans air cleaners that emit a certain amount of ozone.
U.S. Rep. Robert “Bobby” Scott (D-Va.), chair of the education and labor committee, said the federal government typically is not involved in local decisions of what products to buy, although he hopes for more federal guidance.
In the meantime, “these school systems are dealing with contractors providing all kinds of services,” he said, “so you just have to trust them to get the best expert advice on what to do.”
These go-between contractors т and the air cleaner companies themselves т have a stake in the sales. While their names might appear in school board records, their role in selling the device or commission from the deal is seldom made public, KHN found.
A LinkedIn with the logo for one air purifier company, ActivePure Technology, which employs former Trump adviser Dr. Deborah Birx as its chief medical and science adviser, recruited salespeople this way: “Make Tons of Money with this COVID-killing Technology!!” The commission, the post said, is up to $900 per device.
“We have reps [who] made over 6-figures in 1 month selling to 1 school district,” the ad says. “This could be the biggest opportunity you have seen!”
тA Tiny Bit of Ozone’
Schools in New Jersey have a particularly easy time buying air cleaners called Odorox: A state education agency lists them on their , with a large unit selling for more than $5,100. Originally used in home restoration and mold remediation, the devices have become popular in New Jersey schools as the company says its products can .
In Newark, administrators welcomed students back to class last month with more than 3,200 Odorox units, purchased with $7.5 million in federal funds, said Steven Morlino, executive director of Facilities Management for Newark Public Schools.
“I think parents feel pretty comfortable that their children are going to a safe environment,” he said. “And so did the staff.”
Environmental health and air-quality experts, though, are alarmed by the district’s plan.
The Pyure company’s Odorox devices are on California air-quality “potentially hazardous ozone generators sold as air purifiers” and cannot be sold in the state.
A company distributor’s research shows that its Boss XL3 device pumps out parts per billion of ozone, a level that exceeds limits set by California lawmakers for the and the EPA standard for ground-level ozone т a limit set to protect children from the well-documented to developing lungs.
That level exceeds the industry’s self-imposed limit by more than 10 times and is “unacceptable,” according to William Bahnfleth, an architectural engineering professor at Penn State who studies indoor air quality and leads the ASHRAE Epidemic Task Force.
Jean-Francois “JF” Huc, CEO of the Pyure company, pointed out that the study was done in a space smaller than they for such a powerful Odorox device. He cautioned that it was done that way to prove that home-restoration workers could be in the room with the device without violating work-safety rules.
“We provide very stringent operating guidelines around the size of room that our different devices should be put in,” he said.
You can’t see or smell ozone, but lungs treat it like a “foreign invader,” said Michael Jerrett, who has studied its health effects as director of the UCLA Center for Occupational and Environmental Health.
Lung cells mount an immune-like response, which can trigger asthma complications and divert energy from normal lung function, he said. Chronic exposure has been linked to more emergency room visits and can even cause premature death. Once harmed, Jerrett said, children’s lungs may not regain full function.
“Ozone is a very serious public health problem,” Jerrett said.
Newark has some of rates in the state, affecting 1 in 4 kids. Scholars have linked outdoor ozone levels in Newark to ER visits and is the leading cause of school absenteeism there.
Adding ozone into the classroom is “just nightmarish,” Siegel, of the University of Toronto, said.
Morlino said the district plans to monitor ozone levels in each classroom, based on the federal Occupational Safety and Health Administration level for working adults, which is 100 parts per billion.
“In our research of the product,” he said, “we’ve determined it’s within the guidelines the federal government produces.”
While legal for healthy working adults, the work-safety standard should not apply to developing children, said Michael Kleinman, an air-quality researcher at the UC Irvine School of Medicine. “It’s not a good device to be using in the presence of children,” he said.
But the devices are going into schools throughout the state that will not be monitoring ozone levels, acknowledged Dave Matisoff, owner of Bio-Shine, a New Jersey-based distributor of Odorox. He said the main safeguard is informing schools about the appropriate-size should be deployed to, a factor in ozone concentration.
Huc, the CEO, said his team has measured levels of ozone that are higher outdoors in Newark than inside т with his company’s units running.
“There is a tiny bit of ozone that is introduced, but it’s very, very low,” he said. “And you get the benefit of the antimicrobial effect, you get the benefit of reduction of pathogens, which we’ve demonstrated in a number of studies, and you get the reduction of VOC [volatile organic compounds].”
Meanwhile, despite expert concerns, the devices continue to pop up in classrooms and school nurses’ offices across the state, said Allen Barkkume, an industrial hygienist for the New Jersey teachers union.
He doesn’t blame schools for buying them, as they’re a lot less expensive than overhauling ventilation systems. Teachers often push for the devices in their classrooms, he said, as they see them in the nurses’ offices and think it’ll keep them safe. And superintendents are not well-versed in air quality’s complex scientific concepts.
“Nothing sounds better than something that’s cheap, quiet, small and easy to find, and we can stick them in every classroom,” Barkkume said.
Tested in Shoebox, Sold for Classrooms
While New York officials are “” the installation of ionization devices due to “potential negative health effects,” schools across the state of New Jersey are installing ionizing devices.
Ten miles away from Newark in Montclair, New Jersey, parents have been over the new Global Plasma Solutions’ ionizing devices in their children’s classrooms. The company website that emits ions like those “created with energy from rushing water, crashing waves and even sunlight.”
The devices emit positive and negative ions that are meant to help particles clump together, making them easier to filter out. The company says the ions can the viral particles that cause covid-19.
But Justin Klabin, a building developer with a background in indoor air quality and two sons in the district, was not convinced.
He spent hours compiling scientific evidence. He created painstaking picking apart the ionizers’ viability and helped organize a petition signed by dozens of parents warning the school board against the installation.
Even so, the district spent $635,900 on installing ionizers, which would go in classrooms serving . The devices are often installed in ducts, an important consideration, the company Charles Waddell , because the ions that are emitted lose their power after 60 seconds.
But the company’s shoebox study and inflated ion blast numbers that helped sell the product last year leave a potential customer with little sense of how the device would perform in a classroom, Zaatari said.
“It’s a high cost for nothing,” Zaatari said. The company has sued her and another air-quality consultant for criticizing their devices. Of the pending case, Zaatari said it is a David-versus-Goliath situation, but she will not be deterred from speaking on behalf of children.
“Size of the [test] chamber has proved not to play a role in efficacy results but rather ion density,” GPS spokesperson Kevin Boyle said in an email. The company notes by its covid-inactivating that they “may include … higher-than-average ion concentrations.”
He also said the company is proud to meet the ASHRAE “zero ozone” certification.
Glenn Morrison, a professor of environmental science and engineering at the University of North Carolina, reviewed on a device combating the covid virus in the air. The device appears to reduce virus concentrations, he said in an email, but noted it would not be very effective under normal building conditions, outside a test chamber. “A cheap portable HEPA filter would work many times better and have fewer side effects (possibly ozone or other unwanted chemistry),” he wrote.
Other parents joined Klabin’s campaign, including Melanie Robbins, the mom of a kindergartner and a child in pre-K. Armed with her background in nonprofit advocacy, she reached out to experts. She and other parents spoke at local government meetings about their concerns.
In April, the superintendent told parents the school would turn off the devices, but parents say they haven’t turned them all off.
“As far as I understand, the district has relied only on information from GPS, the manufacturer,” Robbins said during a Montclair Board of Education . “This is like only listening to advice from Philip Morris as to whether smoking is safe or not.”
Dan Daniello, of D&B Building Solutions, an HVAC contracting company, defended GPS products during the meeting. He said they are even in the White House, a selling point the company has .
The catch: A GPS contractor installed its ionization technology in the East Wing of the White House after it was purchased in 2018 т before covid emerged, according to GPS’ Boyle. But the company was still using the White House logo as a marketing image on its website when KHN asked the White House about the advertising in April. It was taken down shortly thereafter.
Boyle said GPS was “recently informed that the White House logo may not be used for marketing purposes, and promptly complied.”
The Montclair school district did not respond to requests for comment.
“I want to bang my head against the wall, it’s so black-and-white,” Robbins said. “Admit this is a poor purchase, the district got played.”
Selling тthe Big Kahuna’
Academic air-quality experts agree on what’s best for schools: More outside air pumped into classes, MERV 13 filters in heating systems and portable HEPA filters. The solution is time-tested and effective, they say. Yet as common commodities, like a pair of khaki pants, these items are not widely flogged by a sales force chasing big commissions.
After covid hit, Tony Barron said the companies pitched air purifying technology nonstop to the Kansas district where he worked as a facility manager last fall.
Pressure came from inside the school as well. Teachers sent links for air cleaners they saw on the news. His superintendent had him meet with a friend who sold ionization products. He got constant calls, mail and email from mechanical engineering companies.
The hundreds of phone calls from air cleaner pitches were overwhelming, said Chris Crockett, director of facilities for Turner USD 202 in Kansas City, Kansas. While he wanted to trust the contractors he had worked with, he tested four products before deciding to spend several hundred thousands of dollars.
“Custodial supply companies see the writing on the wall, that there’s a lot of money out there,” he said. “And then a lot of money is going to be spent on HVAC systems.”
ActivePure says on its website that its air purifiers are in . In , the company said they were “sold through a nationwide network of several hundred franchises, 5,000 general contractors/HVAC specialists and thousands of individual distributors.”
Enviro Technology Pros, founded in January, is one company pitching ActivePure to HVAC contractors. , the founders said contractors can make $950 for each air-cleaning device sold, and some dealers can make a month. Citing the bounty of the billions in federal relief, another video touted ready-made campaigns to target school principals directly.
After KHN asked ActivePure for comment, the Enviro Technology Pros YouTube videos about ActivePure were no longer accessible publicly.
ActivePure did not respond to requests for comment but has said its devices are effective and one is validated by the Food and Drug Administration.
An Enviro Technology Pros founder, Rod Norman, told KHN the company was asked to take the posts down by Vollara, a company related to ActivePure. He called sales to schools “the big kahuna.”
Shortly after he spoke with KHN, the website for his own company was taken down.
In an the company had asked: “4000 classrooms protected why not your kids?”
Shoshana Dubnow contributed to this report.
бюЙѓхњДЋУНвюl Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFFтan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/public-health/as-schools-spend-millions-on-air-purifiers-experts-warn-of-overblown-claims-and-harm-to-children/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=1301481&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>